Announcements for Friday, April 10, 1998
The average for Exam III is 59. Not Bad! There
apparently is a problem with 1d)...you have not done that
experiment yet. So we will discuss what to do about that
in class Friday.
The high score on the exam was 94 by Benjamin Flint
(a repeat offender). CONGRATULATIONS
BEN!! He'll get another bumper sticker in class on
Friday.
I'll have new grades up sometime this weekend. Check
here.
Dr. Corinna Czekaj, Editor of the Kirk-Othmer
Encyclopedia on Chemical Technology, at John Wiley, Inc.
has provided me with some information about
trichloroacetic acid (our favorite acid).
Trichloroacetic acid is a specialty
chemical finding application as an
astringent, antiseptic, and polymerization
catalyst. The sodium salt is used as a
herbicide and has antimicrobial activity. It
is produced by the exhaustive chlorination of
acetic acid using Cl2 and P as a
catalyst. About 1,000 metric tons are
produced in the US each year.
Specialty chemicals are compounds
produced in low volumes at high prices
(relative to commodity chemicals) to solve a
particular customer need; most have more than
one use and many grades/property
specification.
One method of preparation of
trichloroacetic acid is use chloral hydrate
and nitric acid. Does anyone know the
structure of chloral hydrate?
What's happening this week.
Experiment #2: Qualitative Cation
Analysis laboratory write-up is due this week
(in laboratory).
This week's laboratory is Experiment #10:
Copper (II) Tartrate Solubility. We will
cover the calculations for this experiment in
class during the week of April 13th. So we
will delay 1 week (due week of April 20th)
turning in this laboratory experiment. Omit
problem #3 of the pre-laboratory questions
when you are doing the pre-laboratory
questions this week. But do do problem #1 and
#2 before coming to laboratory.
Experiment #9: Determination of an
Equilibrium Constant for a Weak Acid. We will
cover the calculations for this experiment in
lecture on Monday, April 6th. The laboratory
write-up for this experiment will be due in
laboratory the week of April 13th. Remember
to do the pre-laboratory questions (#4, 5f
and 5g) before turning in the laboratory.
Problem Set 12 will probably be due on
Monday, April 13th at 1:30 p.m. in class.
Alright everyone..I'm mad and I
won't take it anymore. If you are not careful about
including the charge on all ions when you are writing the
chemical equations which describe how a species behaves
as an acid or a base I will deduct points!!! For example,
NH3 is a base and
when it is added to water the following equation
describes how it behaves as a base;
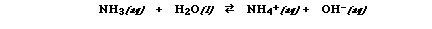
Notice that as a base ammonia
accepts a proton, H+, to become NH4+.
Water on the other hand donates a proton leaving OH-.
Now that's not so hard is it? Remember, reactions
must have a mass balance AND a charge balance or they are
NOT balanced!
You will be responsible for
problems 1 - 6 from Problem Set #12 on Exam III.
New stuff is now
available;
Sample Exam III
Suggested Problems from Chapters 15, 16 and
17.
Pages from Chapter 15, 16 and 17 you are
responsible for on Exam III.
Whoops...I forgot to discuss how we determine the
relative strength of oxy-acids. There are problems on
PS#11 that require you to understand this material. Read
Section 16.10 in BLB for problem 11.8 and 11.9.
Problem Set #12 is ready.
Table 4.1 on page 117 there is a list of the
important strong acids and bases. You should memorize
this table. (NOTE: watch out for H2SO4.)
Currently I have migrated all of the files with the
exception of the LectureNotes to the new server. If you
encounter any problems please contact me via e-mail or
call (47005).
So you love mathematics and can multiply
11 x 16 in your head, or figure out if 112347 is
divisible by 3 in 10 femtoseconds. Then you should
consider the Freshman Math Contest on Saturday April 18,
1998 from 10:00 a.m. to 11:30 a.m. in MS101. Register in
the Math Department (MS401) by April 10th. WIN CASH AND
BOOKS! You must be a freshman, or have fewer than 28
college credits counting towards graduation, excluding AP
credit from High School.
Justin Brooks has done it again. He has
made an Excel
spreadsheet which allows you to enter your grades for
homework, laboratory and exams and it will calculate what
your grade is in class. This is VERY handy and if you
fool around with it you can figure out what you need to
do to get a particular grade. Thanks Justin!
Engineering Student Services is offering
FREE Tutoring to CHEM1515 students. From 6:00 p.m to 8:00
p.m. on Tuesday(s) in Drummond 5. If you need more
information contact Dr. Nichols at CEAT Student Services.
SCHOLARSHIP INFO: Dr. Robert Graalman
(Director of University Scholarships) is organizing the
Phi Kappa Phi Scholarships for this year's awards. He has
application forms for the sophomore $500 awards (for any
OSU student).
Important
Laboratory issues:
How many
of you out there no how to graph data? Using
Excel? Some other software? If you think
you are going to need some help send me an e-mail
or say something to me in class. We may want to
discuss this during a future HELP Session.
This brings up an important point
that I need to make regarding Pre-laboratory
Questions. These questions MUST be completed before
beginning the laboratory. If they are not completed
when the TA checks them 3 points will automatically
be deducted from the total points you earn for that
particular laboratory experiment. Additionally if any
of the points for that experiment are associated with
the pre-laboratory questions you will also loose
those points. I would predict that a student could
loose from 3 to 5 points for NOT completing the
pre-laboratory questions.
Be sure to take your calculator and
your textbook with you to laboratory. Since the first
50 minutes of the laboratory period is
discussion/problem solving you will want to ask
questions about problem sets and or lecture.
Previous Announcements Pages
![]()