 Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index AP Chemistry by Satellite Lectureguide
Student Edition
The Periodic Table
Chapter 5
Objectives
Following your study of this chapter, you should be able to
![]()

![]()
![]()

1. Briefly explain the significance of the atomic number of an element and its position
in the periodic table. Since Mendeleev did not know about atomic numbers, how
did he organize his periodic table?
![]()
2a. In the blank periodic table below identify the areas of the table that contain the
representative elements, transition metals, lanthanides and actinides.
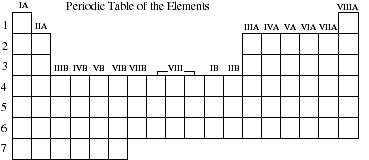
![]()
![]()
![]()
![]()
b) In the blank periodic table below identify the areas of the table that contain the
alkali metals, alkaline earth metals, chalcogens, halogens and noble gases.

![]()
![]()
![]()
![]()
3. In the blank periodic table below identify the areas of the table that contain the
metals, nonmetals and metalloids.

![]()
![]()
![]()
4a. Identify some of the physical properties of the following metals..
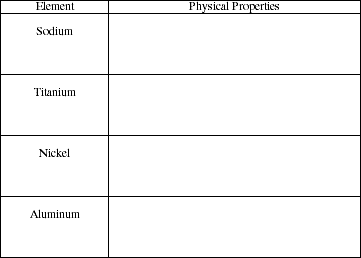
![]()
![]()
4a. continued

b) List five important properties that distinguish metallic elements from nonmetallic
elements.
![]()
c) Explain how the metallic behavior changes going down a group and across a
period.
![]()
d) Identify the physical properties of the following nonmetals.
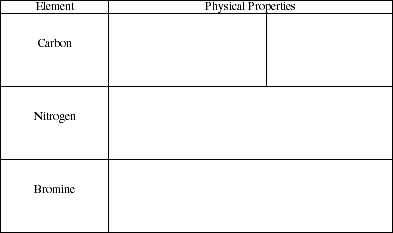
![]()
![]()
4d. (Continued)

e) List five important properties that distinguish nonmetallic elements from metallic
elements.
![]()
f) Explain how the nonmetallic behavior changes going down a group and across a
period.
![]()
5. Correlate the tendency for an element to gain or lose electrons with its position in
the periodic table.
![]()
6. Define oxidation and reduction in terms of electron transfer among reacting
substances.
![]()
![]()
7. Correlate the tendency for an element to be oxidized or reduced with its position in
the periodic table.
![]()
8a. Write the general equation describing single replacement reactions.
![]()
b) Write the products formed when the following substances are mixed.
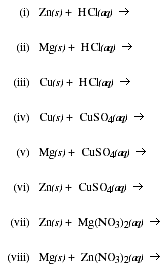
![]()
![]()

9a. Below is table showing the relative activities of several metals.

![]()
![]()
9b) Describe how the activity series is used to predict products of single replacement
reactions.
![]()
c) Write the products formed when the following substances are mixed,
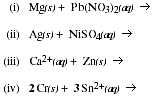
10. What kind (or kinds) of elements are typically found in compounds containing
ions? What kind (or kinds) of elements are typically found in compounds
containing molecules?
![]()
11. Do metal oxides behave as acids, bases, or neutral compounds when added to
water? Do nonmetal oxides behave as acids, bases, or neutral compounds when
added to water?
![]()
12a. Explain why Group IA metals do not occur in nature as uncombined elements.
![]()
![]()
12b. Write the products formed when the following substances are mixed,
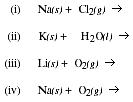
c) Describe the trend in melting point and density which occurs in the Group IA
elements.
![]()
13a. Describe the difference in reactivity of the Group IA and Group IIA elements with
water.
![]()
b) Write the products formed when the following substances are mixed,
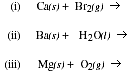
c) Describe the trend in melting point and density which occurs in the Group IIA
elements.
![]()
14. Aluminum is widely used in construction, yet it is ranked as an active metal in the
activity series. Suggest an explanation for this anomaly.
![]()
![]()
15. Describe some of the physical and chemical properties of hydrogen.
![]()
16. Describe some of the physical and chemical properties of the chalcogens.
![]()
17. Describe some of the physical and chemical properties of the halogens.
![]()
18. Describe some of the physical and chemical properties of the noble gases.
![]()
![]()
19a. How would you distinguish between the elements, oxygen, hydrogen, chlorine,
nitrogen and iodine using any combination of physical or chemical properties?
![]()
b) How would you distinguish between the elements, sulfur, sodium, magnesium,
aluminum and bromine using any combination of physical or chemical properties?
![]()
![]()
Problem Set #8
AP Chemistry by Satellite
![]()
ALL work must be shown in all problems for full credit.
PS8.1. Consult the periodic table and arrange the following pure solid elements in order
of increasing electrical conductivity at room temperature: Ca, Sn, S, and Si.
Explain the reason for the order you chose.
![]()
PS8.2. a) Arrange the following series of element in order of increasing metallic
character: Si, Sn, C, Ge, Pb.
![]()
b) Arrange the folllowing series in order of inreasing nonmetallic character:
As, P, Bi, Sb, N
![]()
PS8.3. Write the formula for the compound formed by the combination of each of the
following pairs of elements.
a) magnesium and iodine
b) gallium and oxygen
c) potassium and sulfur
![]()
PS8.4. Which of the following compounds are ionic and which are molecular:
N2O, Na2
O, CaO, CO, P2O5
, Cl2O7
, Fe2O3
? Explain your reasoning.
![]()
![]()
PS8.5. Predict the products and write balanced chemical equations for each of the
following pairs of reactants.
a) Hydrochloric acid and nickel
![]()
b) sulfuric acid and iron
![]()
c) hydrobromic acid and zinc
![]()
d) acetic acid (HC2H3O2) and magnesium
![]()
PS8.6. Based on the acitivitiy series, predict the products of each proposed reaction.
Write a balanced chemical equation for each reaction that occurs.
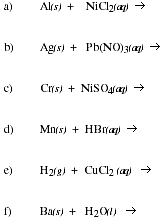
![]()
PS8.7. a) Use the following reactions to prepare an acitvity series for the halogens:
![]()
![]()
![]()
PS8.7b. Relate the positions of the halogens in the eriodic table with their locations
in this activity series.
![]()
c) Predict whether the following reactions will occur.
![]()
PS8.8. Write balanced chemical equations for the reactions that occur when the
following substance are mixed.
a) sodium oxide and water
![]()
b) copper (I) oxide and nitric acid
![]()
c) sulfur trioxide and water
![]()
d) Selenium odioxide and sodium hydroxide
![]()
PS8.9. Write the symbol including the charge for each of the following ions:
a) selinide
b) astinide
c) phosphide
d) cesium ion
![]()
![]()
PS8.10. From the following list of elements select the one that best fits each description:
oxygen
sulfur
lithium
silver
iodine
germanium
aluminum
a) An acive metal that forms a cation with a +1 charge.
b) A grayish-black solid that readily forms a purple vapor.
c) A metal that dissolves in NaOH solution.
d) A metalloid.
e) A yellow nonmetal.
![]()
![]()
Microcomputer software
![]()
Introduction to General Chemistry by Stan Smith, Ruth Chabay and Elizabeth Kean
Drill-and-practice software
$500 (10-disk set)
Falcon Software
P.O Box 200
Wentworth, NH 03282
1-603-764-5788
Diskette #3 Chemical Formulas and Equations
Diskette #6 Chemaze
![]()
Computer Aided Instruction for General Chemistry by William Butler & Raymond Hough
Drill-and-practice software
$40 (4-disk set)
John Wiley & Sons, Inc.
605 3rd Avenue
New York, NY 10158
(this software may not be available)
Diskette #1 Periodic Properties of the Elements
Diskette #2 Nomenclature and Oxidation Numbers
![]()
![]()