 Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index AP Chemistry by Satellite Lectureguide
Student Edition
Chemical Equilibrium
Chapter 14
Objectives
Following your study of this chapter, you should be able to
![]()
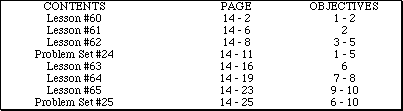
![]()
![]()

![]()
Initially (before any reaction occurs) a 1.00 liter reaction vessel at 25 ºC contains
0.300 moles of NO2 and no nitrogen monoxide or dioxygen. Consider the
following:
a) If 0.100 moles of NO2 react, how many moles of NO and O2 are formed? How
many moles of NO2, NO, and O2 remain after completion of the reaction?
![]()
b) If 0.0873 moles of NO2 react, how many moles of NO and O2 are formed?
How many moles of NO2, NO, and O2 remain after completion of the reaction?
![]()
c) If '2x' moles of NO2 react, how many moles of NO and O2 are formed (in terms
of 'x')? How many moles of NO2, NO, and O2 remain after completion of the
reaction?
![]()
![]()
1. Complete the equation for each reaction demonstrated in lecture and describe your observations.
![]()
Observations:
![]()
![]()
Observations:
![]()
![]()
Observations:
![]()
![]()
![]()
Observations:
![]()
Discuss the differences between the completeness of reactions in a) and b) and in
reactions c) and d).
![]()
2. Complete each of the sections according to the computer experiments performed
in lecture.
![]()
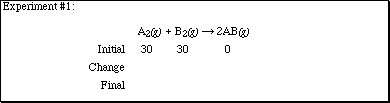
![]()
![]()
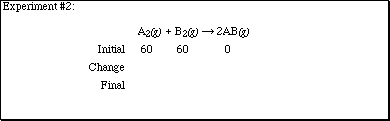
![]()
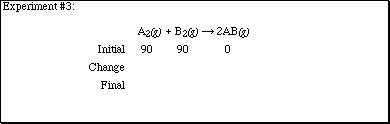
![]()
![]()

2. (Continued)
![]()
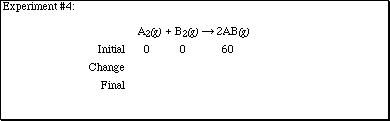
![]()
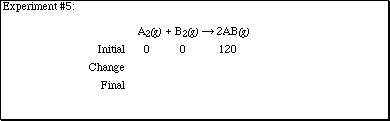
![]()
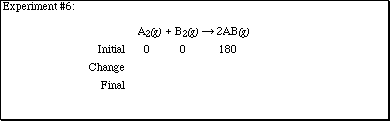
![]()
![]()
![]()
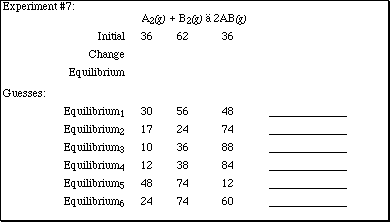
For each of the guesses listed in Experiment #7 above, provide an explanation
why it may be a reasonable answer or not.
![]()
![]()

3. Define the term chemical equilibrium.
![]()
4. Write the equilibrium expression, given the following general equation.
![]()
![]()
Distinguish between the Kp and Kc for the reaction.
![]()
5a. Distinguish between the equilibrium constant and the equilibrium constant
expression for a chemical reaction.
![]()
![]()
b. The following reaction is at equilibrium at a particular temperature
![]()
and the [H2]eq = 0.012 M, [I2]eq = 0.15 M and [HI]eq = 0.30 M. Calculate the magnitude of Kc for the reaction.
![]()
Ans: Kc = 50
c. Using the equilibrium constant calculated in b, calculate the magnitude of the equilibrium constant for the following reactions at the same temperature.
![]()
![]()
Ans: Kc = 0.02
![]()
![]()
![]()
Ans: Kc = 7.07
![]()
![]()
c. The initial concentration of both H2 and I2 is 0.250 M. The reaction occurs as shown below,
![]()
When equilibrium is achieved the concentration of HI is 0.393 M. Calculate the magnitude of Kc for the reaction.
![]()
Ans: Kc = 53
![]()
d. A vessel initially has a partial pressure of NO equal to 0.526 atm and a partial
pressure of Br2 equal to 0.329 atm. At equilibrium the partial pressure of Br2 is
0.203 atm. Calculate Kp for the reaction
![]()
![]()
Ans: Kp = 4.17
![]()
![]()
Problem Set #24
AP Chemistry by Satellite Name___________________________________
![]()
ALL work must be shown in all problems for full credit.
![]()
PS24.1. Given the reaction
![]()
Initially (before any reaction occurs) a 1.00 liter reaction vessel at 400 ºC contains 0.502 moles of O2 and 0.791 moles of NH3 and no water or nitrogen. Consider the following:
a) If 0.0873 moles of O2 react, how many moles of NH3 must react and how many moles of H2O and N2 are formed? How many moles of O2, NH3, H2O and N2 remain after completion of the reaction?
![]()
b) If 0.234 moles of NH3 react, how many moles of O2 must react and how many
moles of H2O and N2 are formed? How many moles of O2, NH3, H2O and N2
remain after completion of the reaction?
![]()
c) If '3x' moles of O2 react, how many moles of NH3 must react and how many
moles of H2O and N2 are formed(in terms of 'x')? How many moles of O2,
NH3, H2O and N2 remain after completion of the reaction?
![]()
d) If 0.875 moles of H2O are formed, how many moles of N2 are formed and how
many moles of O2 and NH3 must react? How many moles of O2, NH3, H2O
and N2 remain after completion of the reaction?
![]()
![]()
PS24.2. Write the equilibrium expression for each of the following chemical equations;
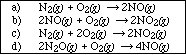
![]()
PS24.3. Equilibrium constants for the following reactions have been determined at 550 ºC:
![]()
Calculate K (at the same temperature) for the commercially important water gas shift reaction
![]()
![]()
PS24.4. Calculate Kc for the reaction
![]()
![]()
if Kc for the reaction
![]()
is 1.3 x 104.
![]()
![]()
PS24.5. A 1.00 liter container initially holds 0.257 moles of NOBr at a given temperature. The reaction which occurs is:
![]()
At equilibrium analysis shows 0.240 moles of NO and 0.120 moles of Br2.
a) Which direction did the reaction proceed to establish (reach) equilibrium?
![]()
b) How many moles of NOBr reacted in order to form 0.240 moles of NO and
0.120 moles of Br2?
![]()
c) How many moles of NOBr remain after equilibrium was established?
![]()
d) What is the magnitude of Kc?
![]()
PS24.6. In a container, the partial pressure of NOCl is initially 0.340 atm at a given
temperature. The reaction which occurs is:
![]()
At equilibrium analysis shows the partial pressure of NO is 0.0916 atm.
a) Which direction did the reaction proceed to establish (reach) equilibrium?
![]()
b) What is the partial pressure of NOCl which reacted in order for the partial
pressure of NO to be 0.0916 atm?
![]()
c) What is the partial pressure of Cl2 at equilibrium?
![]()
d) What is the partial pressure of NOCl at equilibrium?
![]()
e) What is the magnitude of Kp?
![]()
![]()
PS24.7. A 1.00 liter container holds 1.06 moles of H2 and 1.57 moles of CO at a temperature of 162 ºC. At this temperature, the following reaction occurs,
![]()
After equilibrium is established, analysis shows 0.200 moles of CH3OH in the container. Calculate the [CO]eq, [H2]eq and Kc.
![]()
PS24.8. The following reaction,
![]()
occurs at 298K. If 2.00 mol of HI are placed into a 1.00 liter container and permitted to react, at equilibrium it is found that 20.0 % of the HI has decomposed. Calculate Kc and Kp.
![]()
![]()
PS24.9 The equation which describes the preparation of ammonia is:
![]()
A 3.000 L reaction vessel initially contains 0.3000 moles N2 and 0.4500 moles H2. When the reaction is allowed to attain equilibrium at a given temperature, analysis determines 0.09992 M N2. Calculate Kc for the reaction.
![]()
![]()

6a. Setup the ICE table for the following general chemical equation. (Assume the reaction proceeds from left to right to establish equilibrium.)
![]()
![]()
b. The equilibrium constant, Kp, for the reaction
![]()
is 0.0202. If the initial partial pressure of H2 = I2 = 0.350 atm, calculate the equilibrium partial pressures of all species.
![]()
Ans: [H2]eq = [I2]eq = 0.327 atm, [HI]eq = 4.64 x 10-2 atm
![]()
![]()
c. The equilibrium constant, Kc, for the reaction
![]()
is 33.3 at 760 ºC. If 0.400 mol of PCl5 are placed in a 2.00 liter container, calculate the equilibrium concentrations of all species.
![]()
Ans: [PCl3]eq = [Cl2]eq = 0.195 M, [PCl5]eq = 0.005 M
![]()
![]()
d. The equilibrium constant, Kc, for the reaction
![]()
![]()
is 33.3 at 760 ºC. If 0.400 mol of PCl5 and 1.0 mol of Cl2 are placed in a 2.00 liter
container, calculate the equilibrium concentrations of all species.
![]()
Ans: [PCl3]eq = 0.196 M, [Cl2]eq = 0.70 M, [PCl5]eq = 0.004 M
![]()
![]()

7a. State Le Chatelier's principle.
![]()
b. Identify three factors which can affect a reaction at equilibrium. Using examples
discussed in lecture, briefly describe how each factor can affect a reaction at
equilibrium.
![]()
![]()
b. (Continued)
![]()
c. The reaction
![]()
has a DH = -1036 kJ. Given the reaction is at equilibrium, predict the direction the reaction will shift when disrupted by each of the following
![]()
i) the amount of H2O is increased
![]()
ii) the temperature of the reaction is increased
![]()
iii) the volume of the container is decreased
![]()
iv) the amount of H2S is decreased
![]()
![]()
8. Define the non-equilibrium reaction quotient, Q, and explain how it can be used to predict the direction a reaction will proceed to establish equilibrium.
![]()
c. The reaction
![]()
![]()
has been carefully studied at 350 ºC and the Kc is 0.079. Which direction will the
reaction proceed to establish equilibrium under each of the following initial
conditions?
![]()
i) [NOBr]o = 0.100 M : [NO]o = 0 : [Br2]o = 0
![]()
ii) [NOBr]o = 0 : [NO]o = 0.100 M : [Br2]o = 0.100 M
![]()
![]()
iii) [NOBr]o = 0.100 M : [NO]o = 0 : [Br2]o = 0.100 M
![]()
iv) [NOBr]o = 0.100 M : [NO]o = 0.100 M : [Br2]o = 0.100 M
![]()
v) [NOBr]o = 0.200 M : [NO]o = 0.0500 M : [Br2]o = 0.100 M
![]()
![]()

9a. Write the general mathematical equation which relates the equilibrium constant for a chemical reaction to temperature.
![]()
b. Define each of the variables in this equation.
![]()
c. In the reaction
![]()
![]()
DH = -57.2 kJ at 25 ºC. The equilibrium constant, Kp, at this temperature is 8.8.
Calculate Kp at 0 ºC.
![]()
Ans: K1 = 72.9
![]()
![]()
d. In the reaction
![]()
![]()
DH = 26.9 kJ at 25 ºC. The equilibrium constant, Kc, at this temperature is 4.9 x
10-6. Calculate Kc at 100 ºC.
![]()
Ans: K1 = 4.3 x 10-5
![]()
10a. Describe the difference between an equilibrium constant and a rate constant for a
chemical reaction.
![]()
b. How does adding a catalyst to a chemical reaction affect the magnitude of the
equilibrium constant for the reaction?
![]()
![]()
Problem Set #25
AP Chemistry by Satellite Name___________________________________
![]()
ALL work must be shown in all problems for full credit.
![]()
PS25.1. A 0.622 gram quantity of COBr2 is sealed in a glass bulb of 0.100 L volume
and heated to a temperature of 73 ºC. At 73 ºC the COBr2 partially decomposes
according to the equation
![]()
![]()
for which Kc = 0.190. Calculate the concentration of each species at 73 ºC.
![]()
PS25.2. The equilibrium constant, Kp, for the reaction
![]()
is 7.31. Calculate the partial pressure of all species at equilibrium for each of the following original mixtures:
![]()
a) 1.0 atm of CO and 1.0 atm of H2O.
![]()
![]()
b) 1.0 atm of CO, 1.0 atm of H2O and 1.00 atm of H2.
![]()
c) 1.0 atm of H2 and 1.0 atm of CO2.
![]()
![]()
PS25.3. At 1000 K the equilibrium constant, Kc, for the reaction
![]()
is 0.833. Calculate the concentrations of all species at equilibrium when 0.200 moles of NO2 are placed in a 2.00 L container at 1000 K.
![]()
PS25.4. At 25 ºC, 0.560 mol of O2 and 0.20 mol of N2O were placed in a 1.00 liter
vessel and allowed to react according to the equation
![]()
When the system reached equilibrium, the concentration of NO2 was found to
be 0.020 M.
![]()
a) What were the equilibrium concentrations of N2O and O2?
![]()
![]()
PS25.4. (Continued)
![]()
b) What is the value of Kc for this reaction at 25 ºC?
![]()
PS25.5. The reaction
![]()
has been carefully studied at 350 ºC and the Kc is 0.079. Which direction (left-to-right or right-to-left) will the reaction proceed to establish equilibrium under each of the following initial conditions?

PS25.6. Consider the reaction
![]()
for which DHrxn = -1036 kJ. Predict how the [SO2] will change when the equilibrium is disturbed by;
a) Addition of O2
b) Addition of H2O
c) Addition of a catalyst
d) Increase in temperature
e) Decrease in the volume of the reaction container
![]()
![]()
PS25.7. The equilibrium constant, Kp, for the reaction
![]()
is 6.25 at 25 ºC and DHº = 34.4 kJ. Calculate the magnitude of the equilibrium constant at 50 ºC.
![]()
PS25.8. Given the reaction
![]()
A 10.0 liter vessel at 298 K initially contains a sample of XeF4 at 0.750 atm. After the reaction achieves equilibrium, the total pressure in the vessel is 1.95 atm. Calculate Kp from this data.
![]()
![]()
PS25.9. The equilibrium constant, Kc, for the reaction
![]()
is 2.50 x 10-6 at a particular temperature. If the [N2]o = 2.00 M, the [O2]o = 1.00 M and the [NO]o = 0 M, calculate the equilibrium concentration of all species.
![]()
PS25.4. A 0.383 gram quantity of crystalline PCl5 is sealed in a glass bulb of 0.100 L
volume and heated to a temperature of 250 ºC, at which temperature all of the
PCl5 has vaporized. At 250 ºC the PCl5 partially decomposes according to the
equation
![]()
If the [PCl3]eq = 1.4 x 10-2 M, calculate Kc at 250 ºC.
![]()
![]()