 Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index AP Chemistry by Satellite Lectureguide
Student Edition
The Condensed Phases: Liquids and Solids
Chapter 11
Objectives
Following your study of this chapter, you should be able to

![]()
![]()

1. In the spaces below sketch molecular-level models for a substance in the gas, liquid and solid phases.
List observable properties that distinguish each physical state.
![]()
![]()
2. List the initial and final phases for each process. For the enthalpy change, specify whether the process is endothermic (DH positive) or exothermic (DH negative).

![]()
3. Sketch a cooling/heating curve for water in the space below. Label the regions
of the graph identifying each individual phase and phase change.
![]()
![]()
4a. Using the following information, complete the sample problems.

![]()
![]()
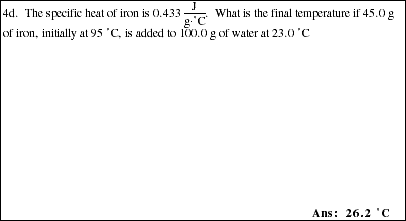
![]()
![]()

5a. Define the term equilibrium vapor pressure.
![]()
b) Use a vapor-pressure table (in your text or some other reference book) to look up
the equilibrium vapor pressure of a sample of water at 28 ºC.
![]()
c) Consider a closed container partially filled with liquid water at 28 ºC. Can the
pressure of water vapor in the gas phase ever exceed the equilibrium vapor
pressure at this temperature? Explain why or why not.
![]()
6a. Order the following liquids from highest vapor to lowest vapor pressure at 25ºC.
water, ethanol, diethyl ether
![]()
![]()
6b. Consider the sketches of four barometers. Barometer i) shows the measurement of atmospheric pressure. Barometer ii) depicts the situation of a sample of water having been injected into the tube. Barometer iii) and iv) depict samples of diethyl ether having been injected into the tubes. All four barometers are at the same temperature.
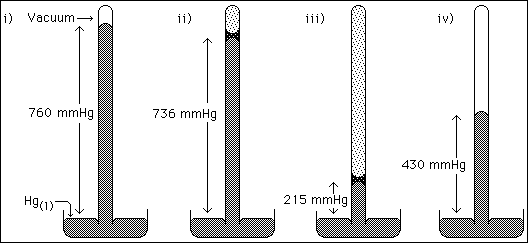
![]()
Why does the height of the mercury column change when liquids are injected?
![]()
c) What is the equilibrium vapor pressure of diethyl ether?
![]()
d) What is the pressure of the diethyl ether vapor in barometer iv)?
![]()
e) Based on your answers in c) and d), what mass of diethyl ether, compared to
that in barometer iii), was originally injected into barometer iv)? (Note: answer
more than, less than, or the same amount as)
![]()
f) Complete barometer iv) by carefully sketching in the space above the mercury
level in the tube, symbols (dots) which correctly represent the phase(s) present.
![]()
![]()
6g. Using your answers for c) through e) explain what happened when the sample of diethyl ether was originally injected into barometer iv).
![]()
7. Using the information on the graph below, explain how a change in temperature
of a liquid effects its vapor pressure?
![]()
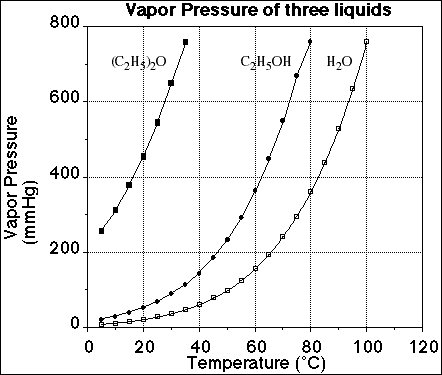
![]()
![]()

8a. Write the Clausius-Clapeyron equation in the space below and define each term.
![]()

![]()
![]()

d) Using the vapor pressure data for acetic acid, CH3COOH(l),

complete the table below and plot ln (Pv) vs. 1/T (K). Use your graph to estimate the heat of vaporization of acetic acid. (Note: ln is the natural log function.)
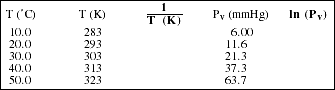
![]()
![]()
8. (continued)
![]()

e. Use the your plot to determine the temperature at which acetic acid has a vapor pressure of 42.4 mmHg.
![]()
f. Use the your plot to determine the vapor pressure of acetic acid at 13.5 ºC.
![]()
![]()
9. Define the terms boiling point, viscosity and surface tension. Describe each property using the kinetic-molecular model.
![]()
![]()
Problem Set #17
AP Chemistry by Satellite
ALL work must be shown in all problems for full credit.
PS17.1. a) How much heat is produced when 75.0 g of steam at 135 ºC is converted to water at 20.0 ºC?
![]()
b) How much heat is required to convert 30.0 g of ice at -10.0 ºC to steam at
105.0 ºC?
![]()
PS17.2. For many years drinking water has been cooled in hot climates by the
evaporation of water from the surface of canvas bags or porous clay pots.
How many grams of water can be cooled from 35 ºC to 20 ºC by the
evaporation of 1.0 g of water?
![]()
![]()
PS17.3. Ethyl alcohol melts at -114 ºC and boils at 78 ºC. The enthalpy of vaporization for ethyl alcohol at 78 ºC is 870 (J/g) and the enthalpy of fusion is 109 (J/g) . If the heat capacity of solid ethyl alcohol is taken to be 0.97 (J/g.ºC), and that for the liquid 2.3 (J/g.ºC), how much heat is required to convert 10.0 g of ethyl alcohol at -120 ºC to the vapor phase at 78 ºC?
![]()
PS17.4. A sample of water vapor in a flask of constant volume exerts a pressure of
530 mm Hg at 100 ºC. The flask is slowly cooled.
a) Assuming no condensation, use the Ideal Gas Law to calculate the pressure of the vapor at 90 ºC; at 80 ºC.
![]()
b) Compare your answers to the equilibrium vapor pressure of water at 90
ºC and at 80 ºC. Will condensation occur at 90 ºC; 80 ºC?
![]()
c) On the basis of your answers in a) and b), predict the pressure exerted by
the water vapor at 90 ºC; at 80 ºC.
![]()
![]()
PS17.5. Consider the following data for the vapor pressure, Pv, of acetic acid, CH3COOH(l) (use graph paper):
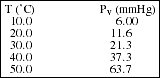
![]()
a) Plot ln (Pv) vs. 1/T and use your graph to estimate the heat of vaporization
of acetic acid.
For parts b) and c) use the Clausius-Clapeyron equation.
b) Determine the temperature of a sample of acetic acid when the vapor
pressure is 42.4 mmHg.
![]()
c) Determine the vapor pressure of acetic acid of a sample of liquid acetic
acid at 13.5 ºC.
![]()
PS17.6. The vapor pressure of bromine at 9 ºC is 113 mm Hg; at 20 ºC it is 184 mm
Hg. Using the Clausius-Clapeyron equation estimate the
a) heat of vaporization of bromine;
![]()
b) vapor pressure of bromine at 50 ºC.
![]()
![]()
PS17.7. The vapor pressure of mercury is 17.3 mm Hg at 200 ºC; its heat of vaporization is 59.4 kJ/mol. Use the Clausius-Clapeyron equation
a) the vapor pressure of mercury at 340 ºC.
![]()
b) the normal boiling point of mercury.
![]()
![]()

10. Sketch the orientations of molecules and/or ions involved in the following intermolecular attractive forces. Include at least one specific example where each attractive force is important. For each one, tell what causes the force and describe its strength relative to the others.
a) ion-dipole forces
![]()
b) dipole-dipole forces
![]()
![]()
10c. London dispersion forces
![]()
d) hydrogen-bonding forces
![]()
![]()
11. Complete the following table.
![]()

![]()
![]()

![]()
![]()

![]()
12. Distinguish between crystalline solids and amorphous solids. Describe how the
difference become apparent when solids of each type melt.
![]()
13. Describe the difference between atomic, molecular, ionic and covalent solids.
Include comparisons of physical properties, such as melting points, as well as
types of intermolecular forces present.
![]()
![]()
14. Sketch the layer structure arrangement for the packing arrangements; simple cubic, body-centered cubic, hexagonal closest-packed and cubic closest-packed.
![]()
![]()
15. Define the term unit cell and sketch the unit cell for simple cubic, body-centered cubic, and face-centered cubic crystals.
![]()
16. Complete the following table.
![]()
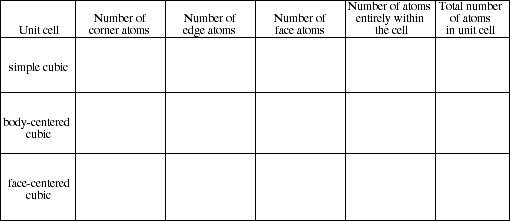
![]()
![]()
17. Distinguish between hexagonal closest-packing and cubic closest-packing in atomic solids. (Sketch the minimum number of layers of circles necessary to show the difference.)
![]()
18. Identify three types of crystal defects and explain how they occur.
![]()
![]()

![]()
19. Sketch a typical phase diagram and label the important features. What
information can be obtained from a pressure-temperature phase diagram?
![]()
![]()
20a. The phase diagram for carbon dioxide is shown below. Complete the diagram by identifying all of the important points, lines and areas. (Hint: There are nine.)
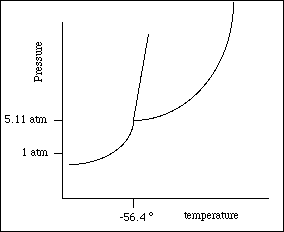
![]()
b) Describe the changes that occur in a sample of carbon dioxide at -52 ºC as
the pressure is increased from 1 atm, holding the temperature constant.

![]()
c) Using the phase diagram, determine the phase of a sample of carbon dioxide
at 5.11 atm and -56.4 ºC?
![]()
![]()
Problem Set #18
AP Chemistry by Satellite
Fall 1991
ALL work must be shown in all problems for full credit.
PS18.1. Indicate what change, if any, should occur in each of the following properties
as a result of an increase in the strength of intermolecular forces:
a) vapor pressure;
b) normal boiling point;
c) normal melting point
d) surface tension;
e) viscosity;
f) heat of fusion;
g) heat of vaporization;
h) molecular weight.
PS18.2. Indicate all the various types of intermolecular attractive forces that may operate in each of the following:
![]()
a) CH3OH(l);
b) Xe(l);
c) H2S(l);
d) ClF(l)
e) Ca(NO3)2(s)
![]()
PS18.3. What are the required structural features for a substantial hydrogen-bonding
contribution to the intermolecular attractive forces?
![]()
PS18.4. List the following compounds in the expected order of increasing energy of
the hydrogen-bonding interaction between molecules:
![]()
H2S; CH3NH2; C6H5OH (phenol).
![]()
![]()
PS18.5. For each of the following pairs of substances predict which will have the higher melting point and indicate why:
(a) CuBr2 > Br2
![]()
(b) CO2 < SiO2
![]()
(c) S < Cr
![]()
(d) CsBr < CaF2
![]()
PS18.6. Indicate the type of crystal (atomic, molecular, metallic, covalent, or ionic)
each of the following would form upon solidification:
![]()
(a) O2
(b) H2S
(c) Ag
(d) KCl
(e) Si
(f) Al2(SO4)3
(g) Ne
(h) SiO2
(i) NH3
(j) MgO
(k) NaOH
(l) CH4 .
![]()
![]()
PS18.7. Sketch the layer structure arrangement for the packing arrangements; simple cubic, body-centered cubic, hexagonal closest-packed and cubic closest- packed.
![]()
PS18.8. Sketch a simple cubic unit cell and explain why only one atom is contained in
a simple cubic unit cell.
![]()
![]()
PS18.9. Using the phase diagram in problem 18.7., determine the physical state of
water at
a) 900 mmHg and 40 ºC
b) 500 mmHg and 30 ºC
c) 300 mmHg and 90 ºC
PS18.10. In the phase diagram for water shown below;

![]()
a) At 400 mmHg what is the approximate temperature needed to convert
water from a solid to a liquid?
![]()
b) What is the approximate pressure at which water changes from a liquid to
a gas at 80 ºC?
![]()
![]()