 Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index AP Chemistry by Satellite Lectureguide
Student Edition
Introduction to Chemistry: Matter and Measurement
Chapter 1
Objectives
Following your study of this chapter, you should be able to
![]()

![]()
![]()

1. List two necessary defining characteristics of matter and illustrate with examples.
![]()
2a. List some physical properties for the substances below that were emphasized during
the Lesson. (Note: You can use your text or the CRC Handbook of Chemistry and
![]()

![]()
![]()
2a. (Continued)

![]()
2b. List some physical properties for aluminum and bromine.
![]()
2c. Describe the chemical change you observed when aluminum is added to bromine.
![]()
2d. Explain why the reaction between aluminum and bromine does not occur
immediately.
![]()
![]()
3. Distinguish between the gas, liquid and solid phase by listing the unique properties of each that are not shared by the others.
![]()
4. Distinguish between pure substances, homogeneous mixtures and heterogeneous
mixtures by listing the unique properties of each that are not shared by the others.
![]()
5. Define the terms atom, element and compound (You may use drawings to support
and clarify your definitions.)
![]()
![]()
6. Distinguish between element and compound by listing the unique properties of each that are not shared by the other.
![]()
7. State the principle postulates of Dalton's atomic theory.
![]()
![]()
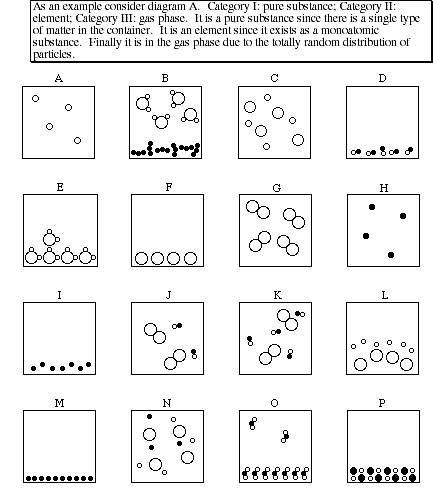
8. Each diagram (A - P) show a sample of substances as viewed at the atomic level. Characterize the contents of the container in terms of each of the following categories:
Category I. Homogeneous mixture, heterogeneous mixture or pure substance
Category II. Element(s), compound(s) or both
Category III. Solid, liquid, gas or combination of phases
![]()
![]()
8. (Continued)
![]()
9/10. Write the names and symbols of the first 36 elements in the periodic table.
![]()
![]()
11. For each of the separation techniques listed below, give an example of how the technique is used to separate the components of a mixture.
a) Decantation
![]()
b) Filtration
![]()
c) Evaporation
![]()
d) Distillation
![]()
e) Paper chromatography
![]()
f) Liquid-solid chromatography
![]()
![]()
12/13. Units for which of the following physical quantities are base units in SI and which are SI-derived units? For each provide the name and symbol (abbreviation) of the SI unit used to express physical quantity.
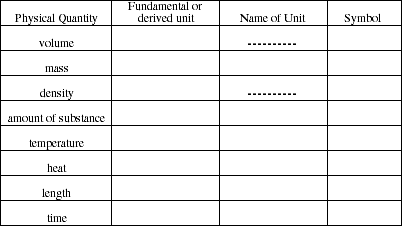
14. Give the name of the prefix and its abbreviation for each of these multipliers.
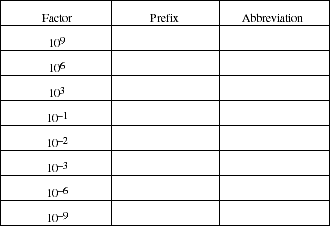
15. Distinguish the meaning of the terms mass and weight.
![]()
![]()
16. Figure I shows a glass cylinder containing four liquids each of different density. Two of the liquids have been identified. A table containing a list of substances and their density (at 25 ºC) has been provided. From the list select a substance for Liquid #1 and Liquid #3. Briefly explain the reason(s) for your selections and for the remaining substances the reason they were not selected.

![]()
![]()
17. The temperature of liquid ethanol was measured several times with two thermometers as the sample was heated from its freezing point to its boiling point. One thermometer was calibrated in degrees Celsius, the other in degrees Fahrenheit. The data collected is shown in the table below.
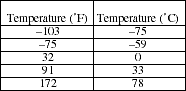
a) Plot the data with ºF on the y-axis and ºC on the x-axis. Determine the mathematical equation relating ºF to ºC.

![]()
b) Use the equation obtained from your graph to calculate the Fahrenheit temperature
corresponding to -40 ºC.
![]()
![]()

18. Distinguish between extensive and intensive properties. Classify each of the physical properties listed as extensive or intensive.
Physical Property Classification
volume
mass
density
temperature
heat
length
time
19a. Compare and contrast accuracy and precision.
![]()

![]()
b) Which set(s) of measurements are precise?
c) Which set(s) of measurements are accurate?
d) Which set(s) of measurements are both precise and accurate?
![]()
![]()
20a. Explain the difference between an exact number and an inexact number.
![]()
b) For each case, indicate whether the result is an exact or inexact number.
(i) The number of apples:
![]()
(ii) The mass of six apples
(iii) The number of seeds in the apples in (i)
(iv) The number of feet in one mile
(v) The number of liters in five quarts
21. List the procedures for determining the number of significant figures in a measured quantity. (Be particularly careful to distinguish between zero as a place-holder and zero as a significant figure.)
![]()
22a. List the procedures for determining significant figures in the result of calculations
involving measured quantities.
![]()
![]()
22b. List the rules for rounding off numbers.
![]()
c) Complete the following calculations and record the result using the correct number
of significant figures.
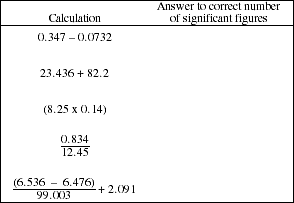
![]()
![]()
23a. List some important unit conversion factors.
![]()

![]()
![]()

![]()
![]()
AP Chemistry by Satellite
Problem Set #1
ALL work must be shown to receive full credit.
PS1.1. Diagram each of the following systems as viewed at the atomic level in the space provided.


![]()
PS1.2. A golden colored cube measures 2.00 cm on each side and has a mass of 154.4 g.
Could the cube be made of gold? Explain your answer using both words and a
mathematical calculation.
![]()
PS1.3. Which temperature of each pair is lower: (Justify each answer with a calculation.)
![]()

![]()
PS1.4. The approximate volume of water contained in the oceans of the world is 3.3 x
108 mi3. Determine the volume in km3.
![]()
![]()
PS1.5. A line of water molecules 2.5 inches long contains 2.5 x 108 molecules. Calculate the diameter of a water molecule in centimeters, angstroms and nanometers.
![]()
PS1.6. The density of water is 1.00 g/mL. What is the weight, in pounds of 1.00 gallon
of water?
![]()
PS1.7a During a flood an average of 1.00 x 103 ft3 of water flowed out of a dam's flood
gate each minute. What volume in ft3 and liters flowed out of the gate in one
hour?
![]()
b) If the flow was constant for one entire day, what mass of water in kilograms was
released from the dam? What weight in pounds?
![]()
![]()
PS1.8. Using a standard linear measuring device (ruler) measure the length and width of this sheet of paper in both inches and centimeters. (Be sure to report the appropriate number of significant figures). Find the area of this page in square centimeters.
![]()
![]()
Microcomputer software
Project SERAPHIM
Dr. John Moore
Department of Chemistry
University of Wisconsin
Madison, WI 53706
AP102 SIGNIFICANT FIGURE DRILL
GRAPH
AP104 ACCURACY AND PRECISION OF LAB MEASUREMENTS DIMENSIONAL ANALYSIS (requires 80-column card)
![]()
![]()
Introduction to General Chemistry by Stan Smith, Ruth Chabay and Elizabeth Kean
Drill-and-practice software
$500 (10-disk set)
Falcon Software
P.O Box 200
Wentworth, NH 03282
1-603-764-5788
Diskette #10 The Metric System
![]()
Computer Aided Instruction for General Chemistry by William Butler & Raymond Hough
Drill-and-practice software
$40 (4-disk set)
John Wiley & Sons, Inc.
605 3rd Avenue
New York, NY 10158
(this software may not be available)
Diskette #1 Introduction: Units and Measurement
![]()
![]()