![]() Pre-lab Questions
Pre-lab Questions
![]() Experiment
Experiment
![]() Post-lab Questions
Post-lab Questions
EXPERIMENT 5: HEAT OF NEUTRALIZATION
![]()
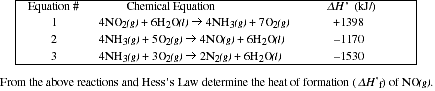
![]()
![]()

List the source you used to find the specific heats.
![]()
![]()
EXPERIMENT 5: HEAT OF NEUTRALIZATION
![]() Top
Top
| EQUIPMENT: | |
| balance...................................1 | labels.....................................4 |
| beaker, 100 mL.........................1 | pencil.....................................1 |
| beaker, 250 mL.........................2 | ring stand................................2 |
| Bunsen burner ..........................1 | rubber band, melting point ............2 |
| calorimeter, (2 Styrofoam cups)......1 | rubber tubing............................1 |
| cardboard,4 inch square ...............3 | spatula ...................................1 |
| graduated cylinder, 50 mL ............2 | styrofoam cup...........................1 |
| iron ring clamp..........................1 | thermometer, ±0.1oC ..................1 |
| matches or lighter........................ | wire gauze...............................1 |
| paper towles......................... several | |
| Apple II Computer .....................1 | |
| adaptor box..............................1 | |
| thermistor probe ........................1 | |
| Project SERAPHIM disk..............1 | |
| printer...............................(optional) |
![]()
WORK IN PAIRS
Computer Set-Up
Examine the adaptor box cord. If it has a round cord with a 9-pin plug, it can be plugged into the matching external port on an Apple IIe, IIc, or IIgs computer. If the cord is a length of ribbon wire with a 16 pin dip adaptor, it must be plugged into an internal port, as described below. Remove the cover of the Apple II computer. Locate a small rectangle on the bottom of the interior, usually near the right rear corner. This is the game port. Notice that the prongs on the adaptor box plug fit the pattern of holes in the game port with the plug oriented either way. One side of the plug has a small white dot or a notch which must be lined up with a similar dot or notch on the game port for the instrument to operate correctly. Plug the adaptor into the game port, being very careful not to bend the prongs. Feed the cord through one of the slits in the back of the computer housing.
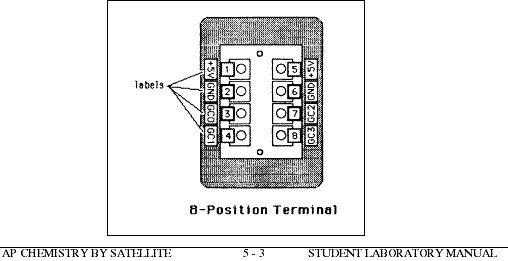
Attach up to four thermistor probes to the adaptor box so that each thermistor has one lead attached to the 5V port and the other lead to one of the game control ports. The leads of the first probe are attached by pressing one of the +5 volt buttons on the adaptor box and inserting one of the lead wires into the hole. Release the button to close the port. Be sure that the wire is firmly connected. Connect the other lead, in the same way, to the connection port labeled GC0. Tug gently on the wires to be sure that they are tightly held. Attach a second thermistor, if desired, to either of the 5 volt ports and GC1. A third thermistor may be connected to either 5 volt port and GC2. The fourth thermistor should be connected to a 5 volt port and GC3. No more than four thermistors can be connected to the computer at any time. If only 1 thermistor is used, it must be connected to GC0.
Place the Project Seraphim disk in the disk drive and turn on the computer and monitor. (Be sure to remove the write protection tabs from the General Laboratory Interfacing diskette before using. If the tabs are not removed, you will not be able to calibrate your thermistor or save data on the diskette.) The disk will boot automatically. Follow the instructions appearing at the bottom of the screen to page through the title and author information. From the first menu, titled "PROGRAM ENTRIES", select option "GENERAL LABORATORY INTERFACING" by pressing the RETURN key when this option is highlighted. Use the arrow keys to move the highlight bar if necessary.
When the "GENERAL LABORATORY INTERFACING" menu appears, select option 1, "THERMISTOR", by typing the numeral "1". Read the descriptions of the functions available and follow the instructions at the bottom of the screen.
Each thermistor must be calibrated before it can be used to measure temperatures. The calibration constants are saved on the floppy disk. As long as the thermistor is connected to the same game control port of the same computer and the same disk is used, re-calibration is not necessary. All of the thermistors attached to the adaptor box must be calibrated in the same temperature baths at the same time. Label each thermistor, using a small piece of tape, with the game control port number.
The response of the thermistor to temperature change is not linear. A minimum of 5 different temperatures are required for calibration, but seven or eight are recommended for best results.
A minimum of 20 kbytes of floppy disk storage must be available on the disk to store temperature and calibration data. Data cannot be saved if there is insufficient storage, and there is no warning! Any data files no longer in use should be deleted from the disk.
![]()
![]()
Before proceeding, ask your instructor if the thermistor probe has already been calibrated. If so, go on to Part A of the procedure.
Heat approximately 100 mL of deionized water in a 250 mL beaker to between 55 and 65 ºC. Pour the water into a Styrofoam cup to minimize cooling.
Using a sharp pencil, make a hole for the thermometer in the center of the cardboard calorimeter top. Make an additional hole near the center for each thermistor to be calibrated. Slide a melting point rubber band over the thermometer and each thermistor to keep them from sliding too far through the cardboard lid. Now slide each one through the lid and position the lid atop the cup. The bottom of the thermometer and thermistor(s) should be about 1 cm above the bottom of the cup and should not touch the side of the cup when the lid is in position.
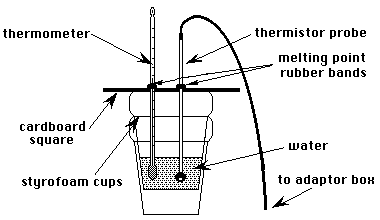
Select option "1", "CALIBRATE THERMISTOR", from the THERMISTOR MAIN MENU by typing the numeral "1". When prompted by the computer, prepare the first water bath. The following temperatures are suggested. Between 50 and 100 mL of water should be used in each case.
| 1) 5-10 ºC | (Place some deionized water in a clean beaker and set the beaker in an ice bath for |
| 15 minutes.) | |
| 2) 22-25 ºC | (Allow water to sit for at least 1 hr to reach room temperature.) |
| 3) 30-34 ºC | (Mix a few mL hottest water with 2) |
| 4) 40-45 ºC | (Add a few more mL of the hottest water to 3) |
| 5) 55-65 ºC | (hottest bath) |
![]()
Place the lid on top of the calorimeter with the thermometer and thermistor(s) immersed in
the water. When the thermometer registers a constant temperature, press the space bar on the
computer keyboard. The computer will now read the thermistors. When a constant value for each
thermistor has been achieved, the computer will prompt you for the Celsius temperature. Type in
the thermometer reading to ±0.1 ºC. If you make an error, use the left arrow key to back up and
re-enter the number. When the temperature has been correctly entered, press the return key and the
![]()
![]()
computer will be ready for the next calibration point. Prepare the next bath and repeat the procedure.
If time permits, calibrate 2 or 3 additional temperatures between 25 and 45 ºC.
When the all of the data has been entered, type "E". The computer will then display the temperature data. Check it for accuracy. Repeat the calibration if the displayed data differ from the recorded temperatures. You may also add more calibration points, if you wish. If the data are satisfactory, the computer will begin calculation of the calibration constants. This will take several minutes. You should use this time to clean the water baths and prepare for the next part of the experiment. If the data are not satisfactory, the computer will tell you so and prompt you to re- calibrate. When the calculation is finished the computer will display the calibration constants and other statistical data. Your instructor may require that you record these values. After this, choose option "2" to save the data on the floppy disk and return to the thermistor main menu.
You are now ready to begin experimental data collection.
![]()
![]()
Part A: Calorimeter Heat Capacity
Use a graduated cylinder to measure 50 mL of room temperature deionized water into a 250 mL beaker. Heat the water slowly (DO NOT BOIL) until the temperature is between 50 and 60 ºC. While the water is heating, use a graduated cylinder to measure 50 mL of deionized water into the calorimeter. Immerse the thermistor probe into the water. Choose option #2 "CONTINUOUS TEMPERATURE SENSING" from the thermistor menu. The calorimeter should not be disturbed for at least 2 min to allow the water to reach thermal equilibrium (constant temperature reading.) Record the temperature to the nearest 0.1 ºC.
TEMPERATURE OF COOL WATER ____________
Transfer the hot water to a styrofoam cup and place a cardboard lid over the top to minimize heat loss. Allow the hot water to stand for a minute or two; remove the thermistor probe from the cool water and place in the hot water. Quickly record its temperature to the nearest 0.1 ºC.
TEMPERATURE OF HOT WATER ____________
Press 'Q' to return to the main menu. Use a new cardboard square for the calorimeter lid. Using a sharp pencil poke a single hole in the center of the square. Put the thermistor probe through the hole so the tip is approximately 1 cm from the bottom of the calorimeter. Use a melting point rubber band to hold the probe in place. Place the lid back on the Styrofoam cup such that the thermistor probe is in contact with the cool water. From the main menu choose "3- SAMPLE TEMPERATURE". When prompted by the computer to enter the time between samples, type "2" and press the return key. The computer will then provide a choice of display formats. Choose option #1, "TABULAR DISPLAY", by typing the numeral "1" followed by RETURN. The final piece of information required is the number of samples. Type "60" followed by RETURN. It is very important to simultaneously initiate data collection by the computer and add the warm water to the cool water in the calorimeter. When everything is ready, one person begins adding the warm water to the calorimeter, and the other person presses any key except "Q" on the computer keyboard. The mixture in the calorimeter should be stirred gently with the thermistor probe. The computer will begin reading the thermistor and displaying the data. The computer will collect 60 data points at 2 s intervals and display this data on the screen. Record the maximum stable temperature in Obs. #3. The computer will 'beep' when all the data has been collected. Press any key to continue.
MAXIMUM TEMPERATURE IN CALORIMETER ____________
![]()
Enter "S" to save the data. Use a file name that has not been used before. The file name
should include both a name (or initials) and an experiment number. Press RETURN.
File name: _________________ (for example, JIG 5A).
After the file is saved you will be returned to the main menu.
![]()
![]()
Part B: HEAT OF NEUTRALIZATION OF HCl(aq) + NaOH(aq)
Dry both the calorimeter and the thermistor probe. Measure 50.0 mL of 1.00 M NaOH and add it to the calorimeter. From the thermistor main menu, select the "2- CONTINUOUS TEMPERATURE SENSING" mode. Place the lid on the calorimeter and measure the temperature of the solution. Now, using a different graduated cylinder measure 50.0 mL of 1.00 M HCl into a clean, dry beaker. After rinsing and drying the thermistor probe, measure the temperature of the HCl solution. NOTE: Allow both solutions to reach equilibrium before recording the beginning temperatures. The temperatures of the two solutions should not differ by more than 0.5 ºC because both should be at room temperature. Press 'Q' to return to the thermistor main menu.
(HAZARDS: Concentrated solutions (6-12 M) of NaOH can cause severe skin burns and permanent damage to eyes. Dilute solutions of NaOH should be treated as hazardous chemicals. Concentrated solutions (6 - 12 M) of HCl can cause severe skin burns and permanent damage to eyes. Inhalation can cause coughing and choking. Dilute solutions of HCl should be treated as hazardous chemicals. If you should come in contact with either solution, rinse the area liberally with water.)
INITIAL TEMPERATURE of HCl ____________
INITIAL TEMPERATURE of NaOH ____________
Now choose "3-SAMPLE TEMPERATURE" from the thermistor main menu. Enter a time interval of 1 s. Press RETURN. Again choose the "TABULAR DISPLAY" format. Enter "60" for the number of samples and press RETURN. Place the thermistor probe and lid over the calorimeter. It is very important to simultaneously initiate data collection by the computer and add HCl solution to the calorimeter. When everything is ready, one person begins adding the HCl to the calorimeter, and the other person presses any key except "Q" on the computer keyboard. The solution in the calorimeter should be stirred gently with the thermistor probe. The data will be displayed on the screen. Record the maximum stable temperature observed in the calorimeter in Obs. #6. Once all the data has appeared on the screen, press any key to continue.
MAXIMUM TEMPERATURE IN CALORIMETER ____________
![]()
Enter "S" to save the data as before using a similar file name changing only the number.
File name: _________________ (for example JIG 5B)
Press RETRUN.
![]()
![]()
Part C: HEAT OF SOLUTION FOR NaOH(s)
Once again choose the "2-CONTINUOUS TEMPERATURE SENSING" mode from the thermistor menu. Place 50.0 mL of room temperature deionized water in a graduated cylinder. Transfer all the water into a 100 mL beaker. Allow the water to reach thermal equilibrium and use the thermistor probe to determine the temperature of the water. Return to the thermistor main menu.
TEMPERATURE OF WATER ____________
![]()
Clean and dry the calorimeter and the thermistor probe. Weigh the calorimeter to the
nearest mg. While at the balance, carefully add 19 to 21 pellets of solid NaOH and record
its mass. You should have approximately 2.00 g of NaOH solid.
(HAZARD: Do not allow NaOH pellets to come in contact with
skin. Solid NaOH is very corrosive to skin tissue. If this
should occur rinse liberally with water.)
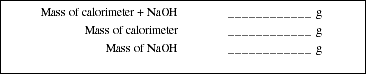
Now choose "3-SAMPLE TEMPERATURE" from the thermistor menu. Enter a time interval of 10 s. Press Return. Again choose the "TABULAR DISPLAY" format. Enter "90" for the number of samples desired. Press RETURN. The extended time interval is required to allow the NaOH pellets to dissolve. It is very important to simultaneously initiate data collection by the computer and add the water to the sodium hydroxide in the calorimeter. When everything is ready, one person begins adding the water to the calorimeter, and the other person presses any key except "Q" on the computer keyboard. The solution in the calorimeter should be stirred gently with the thermistor probe to assist in dissolving the sodium hydroxide. The data will be displayed on the screen. Record the maximum stable temperature observed in the calorimeter in Obs. #9. (Note: If the temperature begins to decrease before the run is completed, check to see if all of the sodium hydroxide pellets have dissolved. If they have, you may stop collecting data by typing "Q." If some of the pellets remain, continue with increased stirring.) Once all the data has been gathered, check to see if all of the sodium hydroxide pellets have dissolved. If they have not all dissolved in this time interval, repeat Part C using a larger number of samples. Press any key to continue.
MAXIMUM TEMPERATURE IN CALORIMETER ____________
![]()
Enter "S" to save data. Enter a file name similar to the previous file name changing only
the number (for example JIG 5C) press RETURN.
File name: _______________
![]()
![]()
Part D: HEAT OF NEUTRALIZATION OF HCl(aq) + NaOH(s)
![]()
Select from the thermistor menu "2-CONTINUOUS TEMPERATURE
SENSING". Place 50.0 mL of 1.00 M HCl in a graduated cylinder. Transfer all the HCl
into a 100 mL beaker. Allow to reach thermal equilibrium, then place the calorimeter lid
and thermistor probe over the beaker. Determine the temperature of the HCl solution.
INITIAL TEMPERATURE of HCl SOLUTION: ____________
![]()
Clean and dry the calorimeter (and thermistor probe). Weigh the calorimeter to the
nearest mg. While at the balance, carefully add 19 to 21 pellets of solid NaOH and record
the mass. It is important that the mass of the NaOH not exceed 2.000 g.
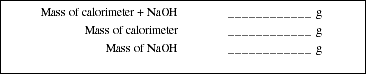
Choose "3-SAMPLE TEMPERATURE" from the thermistor menu Enter a time interval of 10 s, then press RETURN. Choose the "TABULAR DISPLAY" format. The number of samples should be approximately 120. (Once again, if this is not a long enough time interval to completely dissolve the NaOH pellets repeat the procedure with a larger number of samples.) It is very important to simultaneously initiate data collection by the computer and add the HCl solution to the sodium hydroxide in the calorimeter. When everything is ready, one person begins adding the solution to the calorimeter, and the other person presses any key except "Q" on the computer keyboard. The mixture in the calorimeter should be stirred gently with the thermistor probe to assist in dissolving the sodium hydroxide. The data will be displayed on the screen. Record the maximum stable temperature observed in the calorimeter in Obs. #12. (Note: If the temperature begins to decrease before the run is completed, check to see if all of the sodium hydroxide pellets have dissolved. If they have, you may stop collecting data by typing "Q." If some of the pellets remain, continue with increased stirring.) Once all the data has been gathered, check to see if all of the sodium hydroxide pellets have dissolved. If they have not all dissolved in this time interval, repeat Part D using a larger number of samples. Press any key to continue.
MAXIMUM TEMPERATURE IN CALORIMETER ____________
Enter "S" to save the data. Enter a file name similar to all the others, but with a number difference. (for example JIG 5D) press RETURN.
File name: ______________
![]()
![]()
Now that all the data have been collected it must be tabulated for calculations to be made. Take the program disk on which your data has been saved to an Apple microcomputer which is connected to a printer. Boot the disk and repeat the procedure used earlier to get to the thermistor main menu. From the menu select option "4-DISPLAY A FILE". Select option "1" to print the file on the screen, then enter the file name. If this is a file you wish to print, select option "2" to print the file on the printer and reenter the file's name. Continue until you have printed out the four sets of data for parts A, B, C, and D. Clearly label the print-outs of experimental data and turn them in with your report.
On each print-out find the maximum temperature reached inside the calorimeter. Circle or otherwise indicate this value. Sketch a plot of temperature (y-axis) vs. time (x- axis) for each set of data.
Complete the calculation for part A and show it to your instructor. Repeat Part A if you fail to obtain a positive value for the heat capacity of the calorimeter. After you obtain an acceptable value for the heat capacity of the calorimeter, complete the remaining calculations.
![]()
![]()
CALCULATIONS - SHOW ALL WORK Part A: Heat Capacity of the Calorimeter
![]()
HEAT LOST BY WARM WATER = ____________
![]()
Calculate the heat gained by the cool water. Show this calculation and result.
HEAT GAINED BY COOLER WATER = ____________
![]()
Calculate the heat gained by the calorimeter by subtracting heat gained by cooler water
from heat lost by warm water. If you do not obtain a positive number, consult with your
instructor. You may need to repeat the procedure.
HEAT GAINED BY CALORIMETER = ____________
![]()
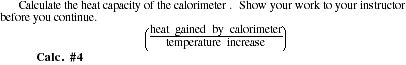
HEAT CAPACITY = ____________
![]()
![]()
Part B: Heat of Reaction HCl(aq) + NaOH(aq)
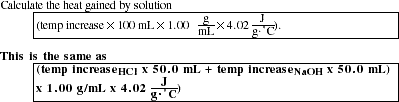
HEAT GAINED BY SOLUTION = ____________
![]()
Calculate the heat gained by calorimeter (temp increase [!] heat capacity of calorimeter).
HEAT GAINED BY CALORIMETER = ____________
![]()
Calculate the total heat released by reaction (solution + calorimeter).
TOTAL HEAT RELEASED BY REACTION = ____________
![]()
Complete the reaction:
HCl(aq) + NaOH(aq)
![]()
Calculate the number of moles of NaCl produced by the reaction in the calorimeter.
Calc. #8
![]()
Calculate the heat of neutralization (heat released per mole of NaCl formed) for the
reaction in the calorimeter. Be sure to include proper units and the appropriate sign for an
exothermic reaction.
![]()
![]()
Part C: Heat of Solution NaOH(s)
![]()
HEAT GAINED BY SOLUTION = ____________
![]()
Calculate the heat gained by calorimeter (temp increase [!] heat capacity of calorimeter).
HEAT GAINED BY CALORIMETER = ____________
![]()
Calculate the total heat released by reaction (solution + calorimeter).
TOTAL HEAT RELEASED BY REACTION = ____________
![]()
Calculate the number of moles of NaOH added to the calorimeter
![]()
Calculate heat released per mole of NaOH added. Be sure to include proper units
the appropriate sign for an exothermic reaction.
HEAT OF SOLUTION = ____________
![]()
![]()
Part D: Heat of Reaction HCl(aq) + NaOH(s)
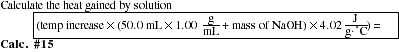
HEAT GAINED BY SOLUTION = ____________
![]()
![]()
Calc. #16
HEAT GAINED BY CALORIMETER = ____________
![]()
Calculate the total heat released by reaction (solution + calorimeter).
TOTAL HEAT RELEASED BY REACTION = ____________
![]()
Complete the reaction:
Rxn. #2
HCl(aq)
![]()
Calculate the number of moles of NaCl produced by the reaction in the calorimeter.
![]()
Calculate heat released per mole of NaCl produced. Be sure to include the
appropriate sign for an exothermic reaction.
HEAT OF REACTION =____________
![]()
![]()
Complete the table below with the important chemical equations that describe the reactions studied in Parts B, C and D and their respective experimental [!]H value.

![]()
Discussion Questions:
Show how the data in the above table support Hess's Law (See Pre-Lab Problem #2). Calculate the percent error for the heat of reaction calculated using Hess's Law compared with the measured value.
![]()
![]()
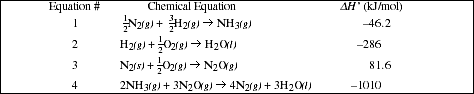
![]()
![]()
(a) 45.0 J.ºC-1
![]()
(b) 0.0 J.ºC-1
![]()
![]()
Contents
![]() Return to
Return to![]() Index of Experiments
Index of Experiments