Advanced Placement Chemistry 1998 Free
Response Questions
6) Answer the following
questions regarding the kinetics of chemical reactions.
(a) The diagram below at right
shows the energy pathway for the reaction
O3
+ NO --> NO2 + O2.
Clearly label the following
directly on the diagram.
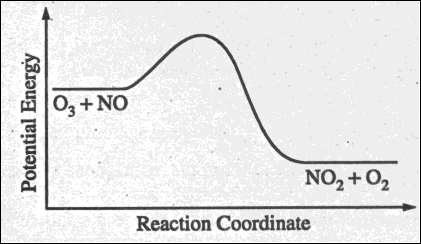
(i) The activation energy
(Ea) for the
forward reaction
(ii) The enthalpy change
(delta H) for the reaction
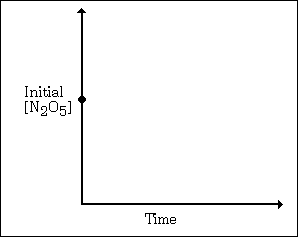
(b) The reaction 2 N2O5
--> 4 NO2 + O2
is first order with respect to N2O5.
(i) Using the axes at
right, complete the graph that represents the change in [N2O5]
over time as the reaction proceeds.
(ii) Describe how the graph
in (i) could be used to find the reaction rate at a given
time, t.
(iii) Considering the rate
law and the graph in (i), describe how the value of the rate
constant, k, could be determined.
(iv) If more N2O5
were added to the reaction mixture at constant temperature,
what would be the effect on the rate constant, k? Explain.
(c) Data for the chemical
reaction 2A --> B + C were collected by measuring the
concentration of A at 10-minute intervals for 80 minutes. The
following graphs were generated from analysis of data.
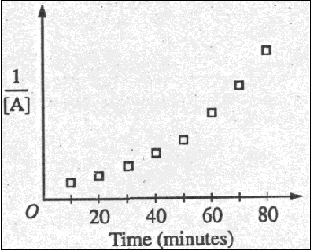
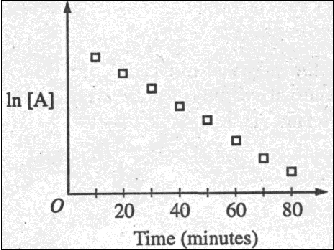
Use the information in the
graphs above to answer the following.
(i) Write the rate-law
expression for the reaction. Justify your answer.
(ii) Describe how to determine
the value of the rate constant for the reaction.
Copyright © 1998 by College Entrance Examination Board and
Educational Testing Service. All rights reserved.