Advanced Placement Chemistry 1995 Free Response Questions
9)
(I) A2 + B2 ---> 2 AB
(II) X2 + Y2 ---> 2
XY
Two reactions are represented above. The potential-energy
diagram for reaction I is shown below. The potential energy of
the reactants in reaction II is also indicated on the diagram.
Reaction II is endothermic, and the activation energy of reaction
I is greater than that of reaction II.
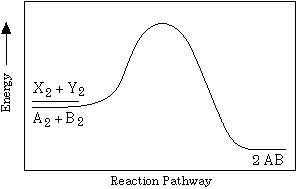
(a) Complete the potential-energy diagram for reaction II on
the graph above.
(b) For reaction I, predict how each of the following is
affected as the temperature is increased by 20°C. Explain the
basis for each prediction.
(i) Rate of reaction
(ii) Heat of reaction
(c) For reaction II, the form of the rate law is rate = k[X2]m[Y2]n.
Briefly describe an experiment that can be conducted in order to
determine the values of m and n in the rate law for the reaction.
(d) From the information given, determine which reaction
initially proceeds at the faster rate under the same conditions
of concentration and temperature. Justify your answer.
Copyright © 1995 by College Entrance Examination Board and
Educational Testing Service. All rights reserved.