The Lewis structure for water;
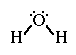
Water is polar. It has two lone-pairs of electrons on the central atom.
Because the oxygen atom is more electronnegative compared to the hydrogen
atom it has a greater attraction to the electrons in the O-H bond. Additionally
the lone-pairs of electrons on oxygen contribute to locating the partial negative
charge on the oxygen atom in the moelcule. Therefore the hydrogen atoms carry
a partical positive charge.
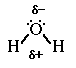
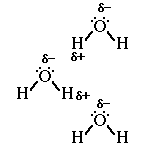
So what happens when several water molecules are located in close proximity?
Can you draw a picture using the Lewis structures depicting the orientation
of three water molecules to each other based on the partial charge carried on
the oxygen and the hydrogen atoms?
Answer.
The adjacent water molecules align themselves so that the partial charges
on adjacen molecules can form an attraction. We refer to the attraction between
the lone-pair of electrons (where there is partial negative charge) on the oxygen
atom of a water molecule and the partial positive charge on the hydrogen atom
as a hydrogen-bond.
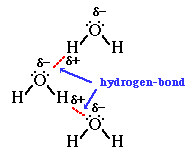
It is important to associate the hydrogen-bond with the intermolecular
attraction between two water molecules, not the O-H covalent bond within (intra)
the water molecule.