Particle |
Charge |
Mass |
electron |
-1.6022 x 10-19 Coulombs |
9.109 x 10-31 kilograms |
proton |
-1.6022 x 10-19 Coulombs |
1.673 x 10-27 kilograms |
neutron |
0 |
1.675 x 10-27 kilograms |
Particle |
Size (meters) |
Atom |
10-10 |
Nucleus |
10-14 |
Proton |
10-15 |
Electron |
10-18 |
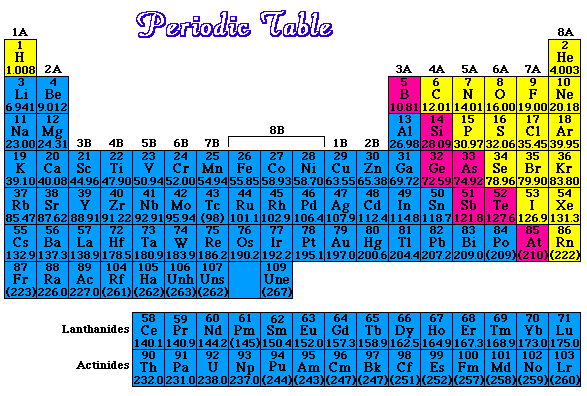
Isotopes of an element are atoms with the same number of protons, but a different number of neutrons.
Symbol |
Atomic Mass (u) |
% Abundance |
1H |
1.0078250 |
99.985% |
2H |
2.0141017 |
0.015% |
The weighted average atomic mass of hydrogen is;weighted average = (atomic mass x fractional abundance)1 + (atomic mass x fractional abundance)2substituting,weighted average = (1.0078250 x 0.99985)1 + (2.0141017 x 0.00015)2 = 1.00794 u |
Isotope |
Relative Mass (u) |
% Abundance |
20Ne |
19.992440 |
90.48 |
21Ne |
20.993846 |
0.27 |
22Ne |
21.991385 |
9.25 |
Complete the following table;
|