According to the chemical equation
4KO2(s) + 2CO2(g) ---> 2K2CO3(s)
+ 3O2(g)
c) how many formula units of potassium carbonate are produced
when six molecules of CO2 react with excess potassium
superoxide?
Answer
According to the balanced chemical equation when two
molecules of carbon dioxide react 2 formula units of potassium
carbonate are formed (assuming excess potassium superoxide).
Carbon dioxide and potassium carbonate always combine in this
ratio according to this equation. We can use this relationship as
a unit conversion factor to convert between potassium superoxide
and dioxygen.
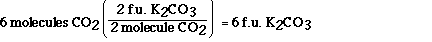
This conversion between carbon dioxide and potassium
carbonate tells us the six formula units of potassium carbonate
are formed when six molecules of carbon dioxide react.