Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index
Name ________________________
AP Chemistry By Satellite
John I. Gelder
Exam IV
May 2, 1991
![]()
INSTRUCTIONS:
1. This examination consists of a total of 8 different pages. The last 2 pages include important mathematical equations and constants, a solubility table, a periodic table, and a table standard reduction potentials. All work should be done in this booklet.
2. PRINT your name, high school, teaching partner's name and today's date now in the space at the top of this sheet. DO NOT SEPARATE THE PAGES.
3. Answer all questions that you can and whenever called for show your work clearly. Your method of solving problems should pattern the approach used in lecture. You do not have to show your work for the multiple choice or short answer questions.
4. No credit will be awarded if your work is not shown in problem 2, 3, 5, 6, 8 and 9. Please circle your final answer!
5. Point values are shown next to the problem number.
6. Budget your time for each of the questions. Some problems may have a low point value yet be very challenging. If you do not recognize the solution to a question quickly, skip it, and return to the question after completing the easier problems.
7. Look through the exam before beginning; plan your work; then begin.
![]()
8. Relax and do well.
![]()

APCBS Exam IV 2
![]()
(12) 1. Complete and balance the following reactions. Identify all product's phases as either (g)as, (l)iquid,
(s)olid or (aq)ueous. Products which are soluble ionic compounds must be written as ions. If no
reaction occurs, write NR.

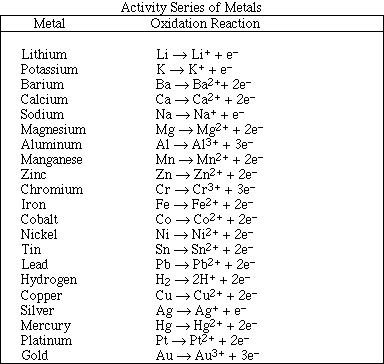
(18) 2. Balance the following oxidation-reduction reactions using the half-reaction method.

identify the oxidizing agent MnO4-
![]() identify the reducing agent HCOOH
identify the reducing agent HCOOH

identify the oxidizing agent HXeO4-
![]() identify the reducing agent HXeO4-
identify the reducing agent HXeO4-
APCBS Exam IV 3
(12) 3. Write the half-reactions and determine Eº for the electrochemical cells as described below, in part b include the overall chemical equation.

b. Pb(s) | Pb2+(aq) || NO3-(aq), H+(aq), NO(g) | Pt(s)

(9) 4. Complete and balance the following reactions. Identify all product's phases as either (g)as, (l)iquid, (s)olid or (aq)ueous. If no reaction occurs, write NR.

Note: I included the half-reactions to indicate the most logical method to determining the products of the reactions. The half-reactions are not required to receive full credit for the problem.
APCBS Exam IV 4
(8) 5. A voltaic cell is constructed inwhich the reaction is,
![]()
If the [Cr3+] = 1.00 M, [Sn4+] = 0.500 M and the [Sn2+] = 0.025, calculate Ecell at 25 ºC.

APCBS Exam IV 5
(8) 6. Calculate [Cr3+] for the reaction in Problem #5 if the [Sn2+] is 1.00 M and [Sn4+] is 1.00 M and the measured cell potential is +0.994 volts.

(8) 7. Write the half-reactions which occur at each electrode when the following solutions are electrolyzed between inert electrodes. If more than one half-reaction can occur at the anode or cathode, write all possible half-reactions and briefly explain which half-reaction will occur.
a. KBr(l)
ANODE:
![]()
CATHODE:
![]()
APCBS Exam IV 6
b. NiCl2(aq)
ANODE:
![]()
Chlorine will be produced at the anode because the overvoltage for the oxidation of water makes the production of chlorine more likely.
CATHODE:
![]()
Ni will plate out at the cathode because the voltage for the reduction of Ni2+(aq) is more positive compared to the voltage for the reduction of water.
(13) 8. When a dilute solution of H2SO4 is electrolyzed, O2(g) is produced at the anode and H2(g) is produced at the cathode.
a. Calculate the coulombs of charge produced when an average current of 8.50 amps passes through the cell for 15.0 minutes.
![]()
b. Calculate the mass of O2 and H2 produced when the cell operates for 15.0 minutes at an average current of 8.50 amps.

APCBS Exam IV 7
8c. The heat of formation of H2O(g) at 25 ºC is -242 kJ/mol. Calculate the heat liberated when the mixture of H2 and O2 in part b. is ignited.

(12) 9. Calculate Eº, DGº, and K for the following reaction