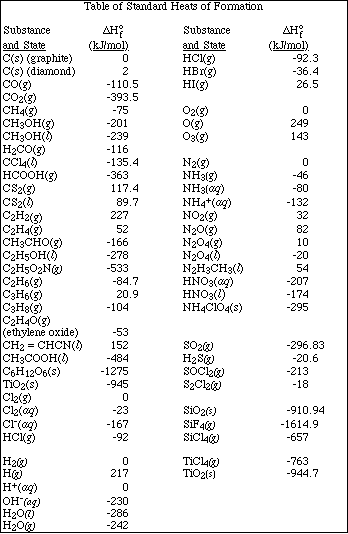
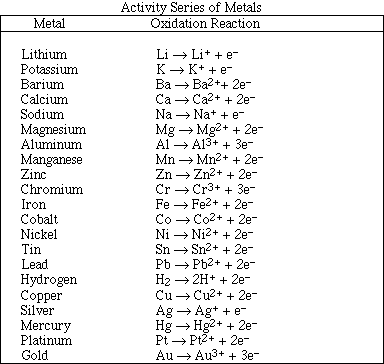
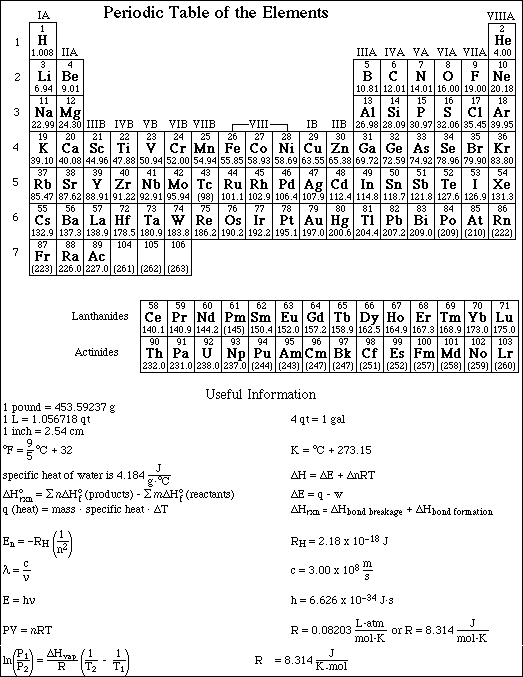Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index
Name ________________________
AP Chemistry By Satellite
John I. Gelder
Exam IV
December 3, 1991
![]()
INSTRUCTIONS:
1. This examination consists of a total of 9 different pages. The last two pages include a periodic table and some useful information and an activity series. All work should be done in this booklet.
2. PRINT your name, high school, teaching partner's name and the date now in the space at the top of this sheet. DO NOT SEPARATE THESE PAGES.
3. Answer all questions that you can and whenever called for show your work clearly. Your method of solving problems should pattern the approach used in lecture. You do not have to show your work for the multiple choice or short answer questions.
4. No credit will be awarded if your work is not shown in problems 4 and 5.
5. Point values are shown next to the problem number.
6. Budget your time for each of the questions. Some problems may have a low point value yet be very challenging. If you do not recognize the solution to a question quickly, skip it, and return to the question after completing the easier problems.
7. Look through the exam before beginning; plan your work; then begin.
![]()
8. Relax and do well.
![]()

APCBS Exam IV KEY PAGE 2
![]()
(9) 1. Write the chemical formula(s) of all the product(s) and balance any three of the following reactions.
Identify all product phases as either (g)as, (l)iquid, (s)olid or (aq)ueous.

(20) 2. Complete the following table

APCBS Exam IV KEY PAGE 3
![]()
(9) 3. Consider the Lewis structure for the compound below:

a) What is the hybridization on each of the carbon atoms?
C1 sp3![]() C2 sp3
C2 sp3 ![]() C3 sp2
C3 sp2 ![]() C4 sp3
C4 sp3 ![]() C5 sp
C5 sp
b) How many sigma (s) and pi (p) bonds are there in this molecule?
13 s-bonds and 3 p-bonds
![]()
(17) 4. (a) What is the volume of a 15.0 g sample of propane (C3H8) gas at STP?
![]()

(b) Assuming that the volume of the container in part (a) remains constant, what would be the pressure of the propane at room temperature (25 ºC)?

(c) If 5.00 g of Ar are added to the container at 25 ºC, assuming no volume change or loss of propane, what is the pressure exerted by the propane and what is the new total pressure?

APCBS Exam IV KEY PAGE 4
![]()
(10) 5. Consider the reaction:
![]()
The hydrogen gas is to be collected over water. If 1.50 g of zinc reacts with excess HCl what volume of hydrogen will be collected? The sample of hydrogen gas is collected at 23.0 ºC. Atmospheric pressure is 745 mmHg when the gas was collected. (Note: the vapor pressure of water is 21.0 mmHg at 23.0 ºC.)

(10) 6. According to the Kinetic Molecular Theory explain how:
(a) kinetic energy (heat) is transferred from a sample at high temperature to a sample at low temperature.
Heat is transferred by collisions between molecules. Molecules at high temperature travel at a greater average speed compared to molecules at a lower temperature. When a molecule with a greater kinetic energy (molecule at a higher temperature) collides with a molecule with a smaller velocity (molecule at a lower temperature) some kinetic energy is transferred from the molecule with greater energy to the molecule of smaller kinetic energy. The result is the average energy of the molecules, initially at a higher temperature, drops while the average energy of the molecules, initially at a lower temperature, increases.
(b) a decrease in the volume of a container of gas at constant temperature causes the pressure to change.
Pressure is a result of collisions of the gas molecules with the walls of the container.
![]()
If the volume of the container decreases, so does the surface area. The same number of molecules with the same average kinetic energy not collide more frequently with the walls of the container and the pressure exerted by the gas increases.
APCBS Exam IV KEY PAGE 5
![]()
(9) Answer either question #7 or #8 on this page. Only the first answer given will be graded.
7. Consider the equipment pictured on the below. How could you use this equipment to determine the value of the absolute zero of temperature? Your answer should address each of the following points:
(A) What measurements would you make in
the laboratory
(B) How would you analyze these
measurements
(C) What assumptions would you have to
make in order to obtain a value for
absolute zero
(D) Which of the gas laws would you verify
in the experiment

![]()
![]()
7. The length of the column of air trapped by the plug of mercury must be measured at
various temperatures. The data is plotted; with temperature on the x-axis and length of the
column of air on the y-axis. The data should produce a straight line. The line through the
room temperature data can be extrapolated to zero on the x-axis. This can be done if assume
the linear relationship is valid outside the range of the experimental data. The temperature at
zero length can be determined. Since a length of zero is the smallest length the temperature
must be the lowest temperature, or absolute zero. This experiment is a verification of
Charles' Law.
APCBS Exam IV KEY PAGE 6
![]()

![]()
8. The equation is M = mRT/PV) . The data which must be measure includes the mass,
temperature, pressure and volume of the gas sample. The mass of a dry erlenmeyer flask is
measured first. Also atmospheric pressure is measured. The erlenmeyer flask, to which a
small amount of the unknown liquid has been added is immersed in a beaker containing boiling
water. At this elevated temperature the sample enters the vapor phase. This is observed until
the point at which the last visible drop of the liquid disappears. At this point the flask is
quickly removed from the hot water and allowed to cool to room temperature. The mass of the
flask and remaining sample is determined. By subtracting the mass of the dry flask the mass
of the unknown sample is obtained. The volume of the flask can be determined by filling the
flask with water and pouring the water into a graduated cylinder. With this data the molar
mass of the gas can be determined. Two assumptions are made; 1) the sample behaves as an
ideal gas when in the vapor phase, and 2) no vapor escapes from the flask after it is removed
from the boiling water.
APCBS Exam IV KEY PAGE 7
![]()
Multiple Choice:
Print the letter (A, B, C, D, E) which corresponds to the answer selected.
9. A ![]() 10. B
10. B ![]() 11. D
11. D ![]() 12. A
12. A
13. E ![]() 14. A
14. A ![]() 15. D
15. D ![]() 16. D
16. D
ONLY THE ANSWERS IN THE AREA ABOVE WILL BE GRADED. Select the most correct answer for each question. Each question is worth 2 points.
9. Which of the following gases would have the greatest density at STP?
A) CO2
B) Ar
C) CH4
D) N2
E) O2
10. Under what conditions do real gases behave most like ideal gases?
A) high temperature and high pressure
B) high temperature and low pressure
C) low temperature and high pressure
D) low temperature and low pressure
E) low temperature at all pressures
11. Consider the manometer shown below. If atmospheric pressure is 0.925 atm, what is the pressure of the gas sample in the syringe?

A) 703 mm Hg
B) 9.5 x 10 -3 atm
C) 710 mm Hg
D) 696 mm Hg
E) 7.2 mm Hg
APCBS Exam IV KEY PAGE 8
12. An equal molar mixture of the four gases listed below is placed in a sealed container. If a pin hole is opened in the container, which gas will be present in the container at the greatest concentration after 10 seconds?
A) SF6
B) Xe
C) Cl2
D) HI
E) The concentration of all gases will be equal.
13. Which of the following substances is not a gas under STP conditions?
A) CO2
B) Cl2
C) Ar
D) CH4
E) Br2
14. Which of the following graphs does not represent the behavior of an ideal gas?

15. Which of the following substances contains bond angles of approximately 109.5º?
![]()
A) SO3
B) NO3-
C) BF3
D) PCl3
E) IF3
![]()
16. The van der Waals equation
![]()
describes the behavior of real gases. Which of the following statements is true about this equation?
A) a is a correction for size of the gas molecules
B) b is a correction for the velocity of the gas molecules
C) b is a correction for the attraction of gas molecules to each other
D) b is a correction for size of the gas molecules
E) the equation approximates the ideal gas equation as a and b each approach a value of 1.00