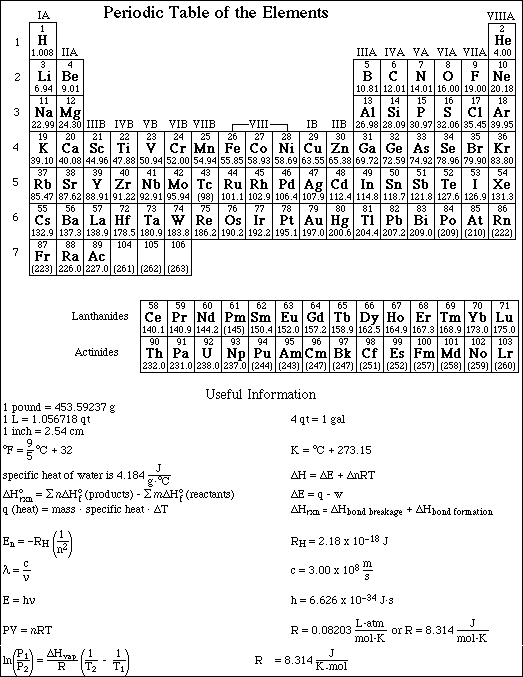Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index
Name ________________________
AP Chemistry By Satellite
John I. Gelder
Exam II
October 10, 1991
![]()
INSTRUCTIONS:
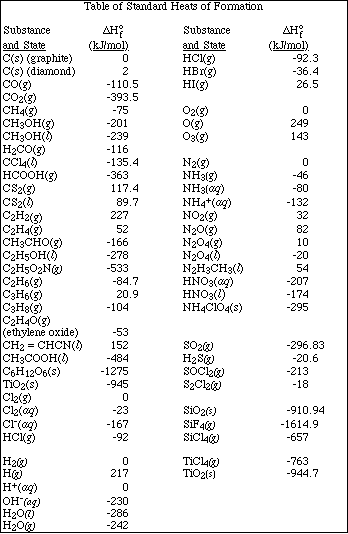
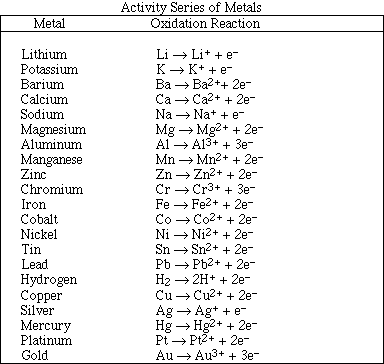
1. This examination consists of a total of 10 different pages. The last three pages include a periodic table and some useful information, a table of standard heats of formation and an activity series. All work should be done in this booklet.
2. PRINT your name, high school, teaching partner's name and the date now in the space at the top of this sheet. DO NOT SEPARATE THESE PAGES.
3. Answer all questions that you can and whenever called for show your work clearly. Your method of solving problems should pattern the approach used in lecture. You do not have to show your work for the multiple choice or short answer questions.
4. No credit will be awarded if your work is not shown in problems 2 - 6.
5. Point values are shown next to the problem number.
6. Budget your time for each of the questions. Some problems may have a low point value yet be very challenging. If you do not recognize the solution to a question quickly, skip it, and return to the question after completing the easier problems.
7. Look through the exam before beginning; plan your work; then begin.
![]()
8. Relax and do well.
![]()

APCBS Exam II KEY PAGE 2
![]()
(9) 1. Write the chemical formula(s) of the product(s) and balance any three of the following five reactions.
Identify all products phases as either (g)as, (l)iquid, (s)olid or (aq)ueous.

![]()
(10) 2. A common procedure for determining the concentration of a solution of base is to neutralize an aliquot of
an acid of known concentration. If a 20.00 mL sample of 0.345 M H2SO4, sulfuric acid, is neutralized
after addition of 15.60 mL of a sodium hydroxide solution, calculate the concentration of the sodium
hydroxide solution. The neutralization reaction is,

If the stoichiometry from the balanced chemical equation is omitted the calculated answer is 0.442 M NaOH.
APCBS Exam II KEY PAGE 3
![]()
(10) 3. Calculate the mass of carbon dioxide produced when 9.00 g of Na2CO3 are added to 50.0 mL of
0.630 M of H3PO4. The reaction which occurs is,

![]()
(10) 4. The final step in the production of nitric acid is described in the reaction below,

APCBS Exam II KEY PAGE 4
![]()
(10) 5. Using the following standard enthalpy of reaction data and Hess' Law calculate the standard heat of
formation for MnO2(s).

(10) 6. Solve ONE of the two problems on this page. (A second problem will not be scored.) Show all important calculations.
![]()
A. 50.0 mL of 1.0 M HNO3 solution and 50.0 mL of 1.0 M NH3 solution are placed in a coffee cup
calorimeter having a heat capacity of 30.3 J/ºC. The density of each solution is 1.00 g/mL and the
specific heat of the solution is 4.20 J/g · ºC. The original temperature was 23.7 ºC and the final
temperature was 24.8 ºC. Determine the heat released in the reaction. How much heat is released per
mole of ammonium nitrate formed?

APCBS Exam II KEY PAGE 5
![]()
B. 0.400 g sample of tristearin (C57H110O6), a common fat, is combusted in a bomb calorimeter containing
3.75 kg of water. The heat capacity of the calorimeter was 532 J/ºC. The measured temperature
increase was 0.938 º C. Determine the heat of combustion per gram of fat in both kJ and kcal (food
calories).
![]()

(12) 8. For each of the following reactions use the activity series to predict the products. If no reaction occurs write NR. Balance the equations and identify the product phases as either (g)as, (l)iquid, (s)olid or (aq)ueous. (Note: Points will be removed if the phases of the products are not provided.)

![]()
(8) 9. Suppose that equal molar amounts of two compounds, lead nitrate and sodium carbonate, react to
produce lead carbonate and sodium nitrate. The reaction occurs in aqueous solution. Lead nitrate,
sodium carbonate and sodium nitrate are all soluble in water and lead carbonate is not.
A) Write a balanced equation including phases to represent the process described.
![]()
APCBS Exam II KEY PAGE 6
![]()
B) If twice as many moles of lead nitrate are added while the amount of sodium carbonate remains constant,
how will the identity of the products change? How will the amount of product change?
The identity of the products will not change.
The amount of product will not change.
![]()
Multiple Choice:
Print the letter (A, B, C, D, E) which corresponds to the answer selected.
10. C ![]() 11. A
11. A ![]() 12. C
12. C
13. B ![]() 14. B
14. B ![]() 15. C
15. C ![]() 16. A
16. A
ONLY THE ANSWERS IN THE AREA ABOVE WILL BE GRADED. Select the most correct answer for each question. Each question is worth 3 points.
10. Which of the following is true about the number of calories per gram in types of food?
A) fat > carbohydrates > protein
B) fat = carbohydrates = protein
C) fat > carbohydrates = protein
D) fat = carbohydrates > protein
11. Consider the reaction:
![]()
Which of the following is true.

12. -25 ºF is equivalent to what temperature?
A) 248 K
B) -13 ºC
C) -32 ºC
D) -46 ºC
E) -77 ºC
![]()
13. The heat of combustion of one mole of solid napthalene (C10H8) to liquid water and gaseous carbon
dioxide is -5162 kJ. Calculate the standard heat of formation of napthalene.
A) 1.02 x 104 kJ/mol
B) 2051 kJ/mol
C) 259 kJ/mol
D) 84 kJ/mol
E) -168 kJ/mol
APCBS Exam II KEY PAGE 7
![]()
14. What volume of 0.225 M Al(NO3)3 is required to prepare 500.0 mL of a solution which is
0.600 M NO3- ion.
A) 1330 mL.
B) 444 mL
C) 188 mL
D) 133 mL
E) there is no sensible answer to this problem
15. Scientists at the University of Oklahoma claim to have discovered a substance, "Fridge-O," which has a specific heat of - 0.653 J/g ºC). If a 20.0 gram block of solid Fridge-O originally at 24.8 ºC absorbs 435 J of heat, what would be its final temperature?
A) 58.1 ºC
B) 33.3 ºC
C) 8.5 ºC
D) -8.5 ºC
E) -33.3 ºC
![]()
16. Which of the following statement concerning Fridge-O is most reasonable?
A) Scientists at Oklahoma State University will probably challenge the claim of the University of
Oklahoma scientists.
C) The scientists at the University of Oklahoma have discovered a substance with ordinary thermal
properties.
D) The scientists at the University of Oklahoma have discovered a substance with unusual thermal properties - only a few other similar substances are known.
E) If the claim of the University of Oklahoma scientists is true, it will not matter much as there would be no market for a substance with Fridge-O's thermal properties.