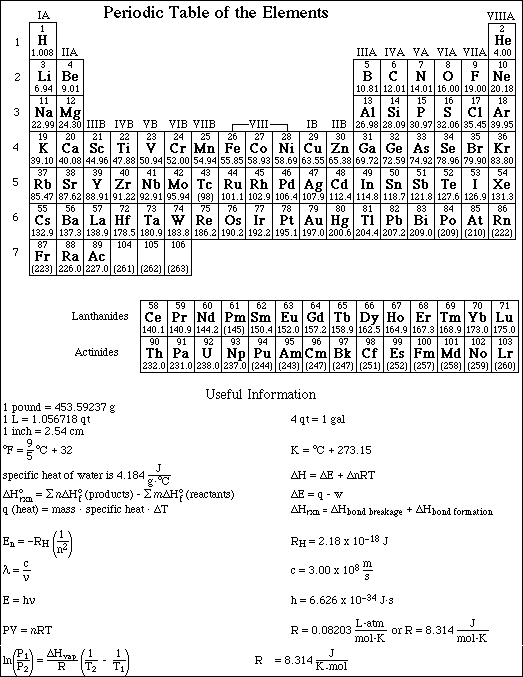Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index
FINAL EXAM PART II
FALL 1991
AP Chemistry by Satellite
Part II of the final examination consists of 37 multiple choice questions. All questions are weighted equally. Page 9 of this exam contains a periodic table and useful information. Additional information which may be required is included with the particular problem. The answer sheet is the last page. Remove it and complete the information listed. Write the letter indicating the most correct answer to each of the 37 questions, clearly and carefully using capital letters on the answer sheet. Your teacher will supply you will additional blank sheets of scratch paper if necessary. You need only turn in the answer sheet to this examination.
Consider the figure for Questions 1 and 2.
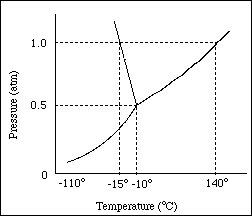
1. The normal boiling point of the substance represented by the phase diagram above is
A. -15 ºC
B. -10 ºC
C. 140 ºC
D. Greater than 140 ºC
E. Not determinable from the diagram
2. For the substance represented in the diagram, which of the phases is most dense and which is least dense at -15 ºC?

APCBS FINAL EXAM PART II PAGE 2
3. The enthalpy of fusion of water is 6.0 kJ/mol and the heat capacity is 75 J/mol ºC. How many kJ of heat would it take to convert 50. g of ice at 0 ºC to liquid water at 22 ºC?
A. 3.8 x 102
B. 21
C. 17
D. 0.46
4. Which of the following substances would have hydrogen bonding as one of its intermolecular forces?
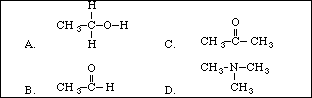
5. Which molecular substance from the following list would have the highest boiling point?
A. N2
B. F2
C. H2
D. Cl2
6. When the actual gas pressure is less than the pressure predicted by the ideal gas law, the explanation lies in the fact that the ideal gas law does NOT include a factor for molecular
A. volume
B. mass
C. velocity
D. attractions
E. shape
7. 25.0 L of hydrogen gas is collected over water at 40 ºC. The total pressure of the sample is 755 mm Hg. At 40 ºC, the vapor pressure of water is 55 mm Hg. What is the mass of the hydrogen gas? (R = 0.08206 L·atm/mol·ºC)
A. 1.81 g
B. 1.95 g
C. 0.896 g
D. 0.967 g
E. 1.12 g
APCBS FINAL EXAM PART II PAGE 3
8. A 2.00 liter sample of nitrogen gas at 27 ºC and 600. mm Hg is heated until it occupies a volume of 5.00 L. If the pressure remains unchanged, the final temperature of the gas is
A. 68 ºC
B. 120. ºC
C. 477 ºC
D. 677 ºC
E. 950. ºC
9. Pi (p) bonding occurs in each of the following species EXCEPT
A. CO2
B. C2H4
C. CN-
D. C6H6
E. CH4
10. Determine the shape and polarity of the PF3 molecule.
A. trigonal pyramidal, polar
B. trigonal pyramidal, nonpolar
C. trigonal planar, polar
D. trigonal planar, nonpolar
11. What is the hybridization and the H - O - C bond angle for the indicated oxygen atom?
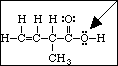
A. sp, 180º
B. sp2, 109º
C. sp3, 109º
D. d2sp3, 90º
12. For which of the following molecules are resonance structures necessary to describe the bonding satisfactorily?
A. H2S
B. SO2
C. CO2
D. OF2
E. PF3
APCBS FINAL EXAM PART II PAGE 4
13. What is the electron configuration of a Co3+ ion?
A. [Ar]4d6
B. [Ar]3d6
C. [Ar]4s23d10
D. [Ar]4s23d4
E. [Ar]4s23d4
14. Which of the following molecules has the shortest bond length?
A. N2
B. O2
C. Cl2
D. Br2
E. I2
15. From the orbital diagrams select an example which demonstrates a ground state atom.
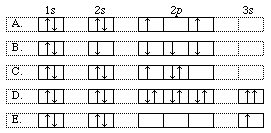
16. One of the outermost electrons in a strontium atom in the ground state can be described by which of the following sets of four quantum numbers?

APCBS FINAL EXAM PART II PAGE 5
Use these responses for Questions 17 - 20.
A. 1s2 2s2 2p6 3s2 3p5
B. 1s2 2s2 2p6 3s2 3p6
C. 1s2 2s2 2p6 3s2 2d10 3p6
D. 1s2 2s2 2p6 3s2 3p6 3d5
E. 1s2 2s2 2p6 3s2 3p6 4s2 3d3
17. An impossible electron configuration.
18. The ground-state configuration for an atom of a transition element.
19. The ground-state configuration of a negative ion of a halogen.
20. The ground-state configuration of a common ion of an alkaline earth element.
21. Which of the following sketches represent orbitals which can have quantum numbers n = 3 and l = 1?
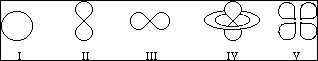
A. V only
B. II and III
C. IV and V
D. I, II and III
E. I, II, III, IV and V
22. Which of the following statements is FALSE?
A. An orbital is a region of space where the probability of finding an electron is
greatest.
B. A Bohr orbit is a circular path of fixed radius and energy which the electron
follows around the nucleus.
C. An emission line in an atomic spectrum is produced when an electron in an
excited state falls to a lower energy level.
D. The energies of photons emitted by excited hydrogen atoms correspond to
the energy differences between Bohr orbits not the energy differences
between quantum mechanical orbitals.
E. The consideration of Heisenberg's Uncertainty Principle and de Broglie's
matter waves account for the difference between the behavior of the electron
in Bohr's model and the quantum mechanical model of the hydrogen atom.
APCBS FINAL EXAM PART II PAGE 6
23. What is the wavelength (in meters) of a photon with an energy of 5.25 x 10-19 J? (c = 3.00 x 108 m/s, h = 6.63 x 10-34 J·s.)
A. 3.79 x 10-7
B. 2.64 x 106
C. 2.38 x 1023
D. 4.21 x 1024
24. Which of the following characteristics is common to elemental sulfur, chlorine, nitrogen, and carbon?
A. They are gaseous elements at room temperature.
B. They form covalent oxides.
C. They have perceptible color at room temperature.
D. They form ionic oxides.
E. They react readily with hydrogen at room temperature.
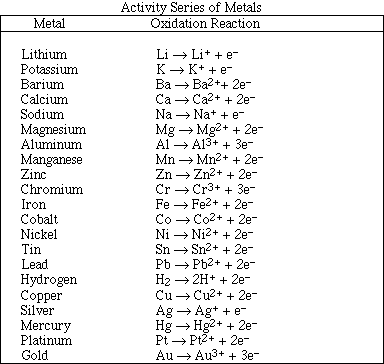
25. Consider the following reactions.
![]()
Which is the correct order of increasing activity for these metals?
A. Ag < Zn < Co
B. Co < Ag < Zn
C. Co < Zn < Ag
D. Ag < Co < Zn
![]()
![]()
What is the standard heat of formation of methane, DHfº CH4 (g) , as calculated
form the data above?
A. -210.0 kJ/mol
B. -107.5 kJ/mol
C. -75.8 kJ/mol
D. 75.8 kJ/mol
E. 210.0 kJ/mol
APCBS FINAL EXAM PART II PAGE 7
27. Consider the reaction:
![]()
Which of the following is true?

28. When 72 g of a metal at 97.0 ºC is added to 100 g of water at 25.0 ºC, the final temperature is found to be 29.1 ºC. What is the specific heat of the metal? (Specific heat of water is 4.184 J/g·ºC.)
A. 0.46
B. 2.8
C. 0.35
D. 2.0
E. 24
![]()
29. How many moles of O2 are needed to produce 14.2 grams of P4O10 from excess
P? (molecular weight P4O10 = 284)
A. 0.0500 mole
B. 0.0625 mole
C. 0.125 mole
D. 0.250 mole
E. 0.500 mole
![]()
30. When 70. mL of 3.0 M Na2CO3 is added to 30. mL of 1.0 M NaHCO3, the
resulting concentration of Na+ is
A. 2.0 M
B. 2.4 M
C. 4.0 M
D. 4.5 M
E. 7.0 M
31. A 27.0 g sample of an unknown hydrocarbon was burned in excess oxygen to form 88.0 g of carbon dioxide and 27.0 grams of water. What is a possible molecular formula for the hydrocarbon?
A. CH4
B. C2H2
C. C4H3
D. C4H6
E. C4H10
APCBS FINAL EXAM PART II PAGE 8
32. How many grams of calcium nitrate Ca(NO3)2 contains 24 grams of oxygen atoms?
A. 164 g
B. 96 g
C. 62 g
D. 50 g
E. 41 g
33. Which of the following compounds is named incorrectly?
A. HCl (g) hydrogen chloride
B. AlCl3 aluminum chloride
C. NCl3 nitrogen chloride
D. CH4 methane
E. CuCl2 copper (II) chloride
34. A radioisotope of carbon undergoes beta decay. Which of the following is a correct product from the decay of:
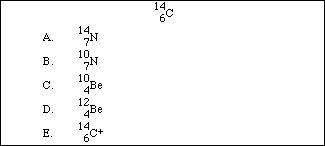
35. A measured mass of an unreactive metal was dropped into a small graduated cylinder half filled with water. The following measurements were made. Mass of metal = 19.611 grams volume of water before addition of metal = 12.4 milliliters volume of water after addition of metal = 14.9 milliliters The density of the metal should be reported as
A. 7.84444 grams per mL
B. 7.844 grams per mL
C. 7.84 grams per mL
D. 7.8 grams per mL
E. 8 grams per mL
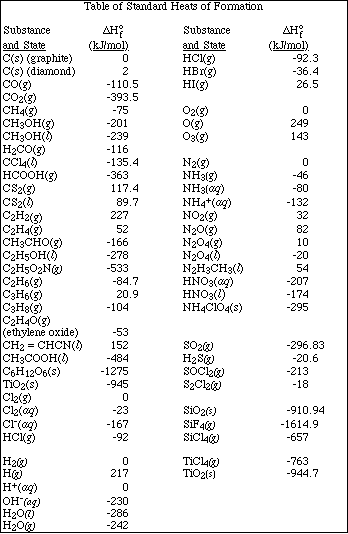
APCBS FINAL EXAM PART II PAGE 9
36. How many significant figures should be retained in the result of the following calculation?
![]()
A. 1
B. 2
C. 3
D. 4
E. 5
37. The table shown below lists the naturally occurring isotopes of element X. Use this data to calculate the average atomic weight of element X.

A. 17.20 amu
B. 16.90 amu
C. 17.65 amu
D. 17.11 amu
|
||||||||||
|
1. 2. 3. 4. 5. 6. 7. 8. 9. |
C D B A D D A C E A C B |
13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. |
B A A E C E B B B D A B |
25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. |
D C C C D D D E C A D B A |
|||||
|
10. 11. 12. |
||||||||||