Go to Main Index
Go to Main Index Go to Main Index
Go to Main Index
FINAL EXAM PART I
Spring 1992
AP Chemistry by Satellite
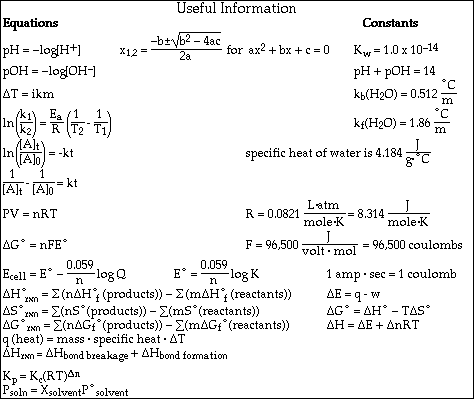
The method used and steps involved in arriving at your answers must be shown clearly. This is important since you may obtain partial credit for doing so, while you will receive little or no credit if you do not. The last page contains a periodic table and important mathematical equations and constants.
PART A: Solve the following problem. (25 percent)
![]()
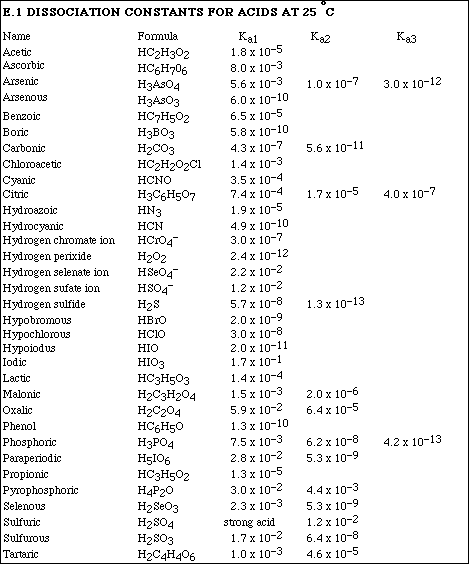
1. Benzoic acid, HC7H5O2, is a weak acid and has an equilibrium constant, Ka, equal
to 6.5 x 10-5 at 25 ºC. A 0.500-liter sample of a 0.150 M solution of the acid is
prepared.
![]()
(a) Write the equilibrium expression for the dissociation of benzoic acid.
![]()
(b) Calculate the pH of this solution.

APCBS FINAL EXAM PART I PAGE 2
(c) To 0.250-liter of this solution 3.20 g of KC7H5O2, is added. The salt dissolves completely. Calculate the pH of the resulting solution at 25 ºC assuming the volume of the solution remains unchanged.

(d) To the remaining 0.250-liter of the original solution, 0.0200 moles of NaOH is added. Calculate the pH of the resulting solution at 25 ºC assuming the volume of the solution remains unchanged.
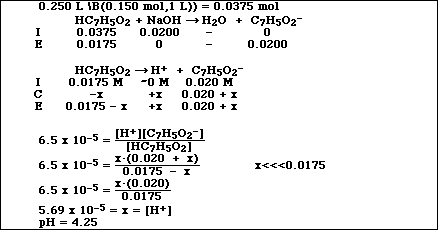
APCBS FINAL EXAM PART I PAGE 3
PART B: Choose ONE of the two problems in this part. (A second problem will not be scored.) (25 percent)
2. The following initial rate data was collected for the reaction at constant temperature;

(a) Determine the reaction order for Ce4+(aq) and Fe2+(aq) .


(b) Write the specific rate law for the reaction?
rate = k·[Ce4+] 1·[Fe2+] 1
(c) Determine the rate constant for the reaction (include units).

(d) Calculate the initial rate if the [Fe2+]0 = 0.250 M and [Ce4+]0 = 0.250 M.
rate = 1.01 x 103 M-1 · s-1·[0.250]1·[0.250]1 = 63.0 M/s
APCBS FINAL EXAM PART I PAGE 4
3. Tungsten(VI) oxide can be reduced to tungsten according to the following equation,

(a) Calculate DHº, DSº, and DGº for the reaction.

(b) Briefly explain the meaning of the sign of each of the thermodynamic values calculated in (a).
DHº = + means the reaction is endothermic
DSº = + means the products are more disorder than the reactants
DGº = + means the reaction is nonspontaneous at 298 K
(c) Calculate the temperature at which the above reaction is spontaneous.

APCBS FINAL EXAM PART I PAGE 5
PART C: Answer FIVE of the eight options in this part. (Answers to more than five options will not be scored.) (20 percent)
4. Write the appropriate formulas to show the reactants and products for the following. All solution are aqueous. For each reactant and product indicate the phase as (s)olid, (l)iquid, (g)as or (aq)eous. Balance the equation. In all cases a reaction occurs.

APCBS FINAL EXAM PART I PAGE 6
PART D: Select THREE of the following five topics. (Additional topics will not be scored.) (30 percent)
5. Draw a diagram of the cell in which the following reaction occurs.
![]()
Label the anode and cathode, the anode and cathode electrode material, the half- reaction at each electrode, the ions in the anode and cathode compartments and salt bridge, the direction of electron flow and the direction of ion movement, and calculate Eº.

6a) Describe how you would determine the magnitude of the equilibrium constant for an unknown monoprotic weak acid of known concentration. You may assume you have access to any standard laboratory equipment and instruments, and any standard reagents.
Measure the pH of the solution using a pH meter. From the
magnitude of the pH and initial concentration of the weak acid the
equilibrium constant can be determined using the following logic.
Assume the formula of the weak acid is HA, then

After measuring the pH the [H+] can be determined using the
equation;
(10-pH) = [H+ ]
According to the ICE table above, the [A-] is equal to the [H+]. The
[HA] is equal to the initial concentration of the weak acid minus the [H+].
The equilibrium constant can then be calculated from the equilibrium
expression,
![]()
APCBS FINAL EXAM PART I PAGE 7
7. In the equation below, the forward reaction is first order in both CO and Br2 and
the reverse reaction is first order in COBr2.
![]()
a) Suppose that 2 moles of CO and 1 mole of Br2 are mixed in a closed container at constant temperature. Draw a graph that shows how the concentrations of CO, Br2 and COBr2 change with time until after equilibrium has been firmly established.

b) Define Le Chatelier's principle.
If a chemical reaction at equilibrium is subjected to a change in
conditions that displaces it from equilibrium, then the reaction
proceeds toward a new equilibrium state in the direction that
offsets the change in conditions.
c) If the above reaction is at equilibrium describe three different conditions which would cause the position of the equilibrium to shift and indicate which direction the reaction would shift to re-establish equilibrium.
1. Concentration of a reactant or product
Addition of CO or Br2 will cause the reaction to shift towards the
product COBr2 increasing its concentration.
2. Reaction volume or pressure
If the volume of the container, inwhich the reaction is occurring, is
increased the reaction will shift toward the reactants.
3. Temperature
There is not enough information to know how the reaction will
shift with a change in temperature.
3. Concentration of product
Addition of COBr2 will cause the reaction to shift towards the
reactants CO and Br2 increasing their concentration.
APCBS FINAL EXAM PART I PAGE 8
8. Arsenic acid, H3AsO4, is a triprotic acid.
a) Draw a correct Lewis structure for arsenic acid.

b) Write the three dissociations equations for the successive loss of hydrogen ion.
![]()
c) Explain the general trend in the magnitude of the equilibrium constant for the three equations.
The magnitude of the equilibrium constants get smaller because it becomes more difficult to remove a proton as the charge on the conjugate base becomes more negative.
d) Which of the three dissociation equations controls the pH of an aqueous solution of H3AsO4? Briefly explain why.
The first equation will control the pH of an aqueous solution of this acid. The first dissociation has the largest magnitude and will produce the most hydrogen ion. While the other two dissociations will occur to a small extent they do not increase the concentration of hydrogen ion apprecibly.
APCBS FINAL EXAM PART I PAGE 9
9. When crystals of barium hydroxide, Ba(OH)2 · 8H2O, are mixed with crystals of ammonium chloride, NH4Cl, at room temperature in an open container, the mixture liquifies, the temperature drops dramatically, and the odor of ammonia is detected. The reaction that occurs is,
![]()
a) Indicate how the enthalpy, the entropy and the free energy of this system change as the reaction occurs. Explain your predictions.
DHº is + because the temperature of the system drops.
DSº is - because the products are more disordered compared to the
reactants.
DGº is - because the reaction occurs when the reactants are
combined.
b) If the beaker in which the reaction is taking place is put on a block of wet wood, the water on the wood immediately freezes and the beaker adheres to the wood. Yet the water inside the beaker, formed as the reaction proceeds, does not freeze even though the temperature of the reaction mixture drops to -15 ºC. Explain these observations.
The aqueous solution contained in the beaker does not freeze even though the temperature is below 0ºC because of the presences of the ions in solution. The ions depress the freezing point of the water in the beaker to a value well below the final temperature of the mixture. The water placed on the board was pure water, which freezes at 0ºC.