CAUTION: Use appropriate safety guidelines in performing demonstrations. Only brief procedural steps are described here. Some of these demonstrations can be used as student activities
.
Demonstration 1: 2NO2(g)
N2O4(g) and Temperature
Purpose
To demonstrate the effect of temperature on a system at equilibrium.
Part A. Appearance of Glass Samples
Materials
Hot water bath Ice water bath 3 NO 2 (g)-filled tubes Dry ice/acetone bath (optional) Overhead projector
Safety
No special precautions are necessary other than those given in the directions.
Procedure
Obtain NO 2 /N 2 O 4 (nitrogen dioxide/dinitrogen tetroxide) gas tubes from an educational scientific supply house (e.g. ,Temperature Equilibrium Tubes, Sargent-Welch #4426). Place one tube into a beaker of hot water 70 to 80 °C, one tube into a beaker of ice water (alternative: a dry ice/acetone bath; it will turn the gas colorless), and leave one tube at room temperature. Place all three tubes on an overhead projector and ask students to make observations. Write the equilibrium equation on the board:
Part B. Density of Glass Samples
Materials
2 Different glass bead samples
Balance Graduated cylinder, 10-mL
Water, 100 mL
<
Procedure
- Add 2-3 mL of water to the graduated cylinder. Read the volume to the nearest 0.01 mL. Determine the mass. Record both the volume and mass.
- Add enough glass beads from one sample to the cylinder to increase the volume by about 5 mL. The additional glass beads should be submerged. Read the new volume to the nearest 0.01 mL and record. Determine the mass of this new combination and record.
- Determine the density of this glass sample and record results.
- In order to be sure your density is reliable, add a few more beads to this same cylinder. Record the final volume and determine the new mass.
- Determine the density of this second trial by subtracting this final volume from the initial volume and by subtracting the final mass from the initial mass. Record results. Take an average of the two densities and record this average on the data sheet.
- Repeat Steps 1-5 for the other glass sample.
Demonstration 2: Refractive Index of Glass
Refractive index is a measure of the bending of light as it passes from air into a solid or liquid. Determination of the refractive index of glass provides information about the common origin (or lack thereof) of two glass samples.
In the immersion method a glass sample is added to a pure liquid or liquid mixture of known refractive index. If the glass and liquid have different refractive indices, a colored boundary can be seen with the unaided eye. If they have the same index of refraction, the boundary will disappear and the specimen will be practically invisible.
Materials
Several fragments of Pyrex` glass
Several fragments of common (flint) glass
Baby oil or vegetable oil
Test-tubes
Safety
Sharp glass may cause cuts and should be handled with care.
Procedure
- Fill three test-tubes about half-full of oil.
- Put some of the Pyrex` fragments in one test-tube, some common glass fragments in another, and a mixture of Pyrex` and common fragments in the third.
- Stopper the three test-tubes. Observe.
- The Pyrex` glass will be nearly invisible when covered with the oil, while the flint glass will remain visible. The Pyrex` glass has an index of refraction (1.47) that nearly matches the index of refraction of the oil, whereas flint glass has an index of refraction of 1.60.
- Discussion. Many crime laboratories have a standard set of liquids of varying refractive index over a wide range and in small increments (typically 0.05). These so-called Cargille liquids allow the forensic specialist to determine the refractive index of transparent samples to an uncertainty of ±0.05 by the immersion method.
Demonstration 3: Analysis of Fingerprints
Fingerprints are the most reliable means of identifying suspects. No
two human fingerprints are exactly alike, and fingerprints do not change
naturally. Fingerprints are classified into three main types: loop, arch,
and whorl (see Figure 8). There is no set number of points of similarity
used by experts when comparing fingerprints for identification. However,
many latent print examiners often use seven points as a minimum. 
Figure 8. Types of fingerprints.
Part A. Obtaining a Fingerprint
Materials
Black ink stamp pad
Tissue paper
4 x 4 cm Card (cut from a 3 x 5 inch file card)
Tweezers
Safety
Wash hands thoroughly to remove the black ink.
Procedure
- Handle the card only on the edges.
- Place your right thumb on the black ink pad and then place your thumb print in the middle of the card.
- Examine the print and identify it by main type.
- Compare with other students in the class who have the same main type to determine if there are distinguishing features in the thumb prints.
Part B. Dusting for and Lifting Prints from a Smooth, Nonporous Surface
Materials
Dusting brush (one for each color of powder)
Dusting powders (aluminum and carbon black).
2 Beakers, 150-mL
Newspaper
2-inch wide Cellophane tape
Index card
Magnifying glass
Safety
Take care not to scatter dusting powders. Some people are sensitive to the airborne particulates.
Procedure
- Grab a 150-mL beaker so that your thumb print is left on the beaker.
- Obtain a brush for dusting the print. Make sure it is clean and the bristles are separated from each other.
- Place a small amount of the dusting powder in a labeled beaker. Dip the brush in the powder and lightly dust the area containing the print. After the entire print is developed, remove the excess powder by gently brushing it away. Be careful not to destroy the print with too hard a brush stroke.
- To lift the print from the 150-mL beaker to the index card, unroll 9 cm of tape. Bend the tape strip in the form of a U so that the sticky side is facing the beaker. Place the point of the fold directly on the print. Gently place the rest of the stripe onto the beaker. The print can be removed by pullinguponthe rollendofthe tapeandthenplacing itonthefingerprint card in the same manner as the tape was placed over the latent print. Make sure the tape is secure. Cut off the excess tape.
- Observe the print under the magnifying glass and compare it with your right thumb print from Part A.
Part C. Using Ninhydrin to Develop a Print on Paper
Ninhydrin reacts with amino acids in the perspiration on a fingerprint to form a purple compound.The reactionofninhydrinwith anaminoacidmay berepresentedasfollows:

The purple-colored substance is formed by the reaction of some of the ninhydrin with its reduction product, hydrindantin, and ammonia, which is formed as a reaction intermediate.
Safety
Wear plastic gloves when handling ninhydrin since it will stain skin. Ethanol is volatile and flammable. Keep the solution away from open flames.
Materials
4" x 5" Sheet of white paper containing your right thumb print
Ninhydrin solution (0.3 g ninhydrin in 100 mL ethanol)
Forceps
Plastic gloves
Brush or cotton wads
Magnifying glass
Concentrated ammonia or steam iron
Procedure
- Tape the top of the exhibit (white sheet of paper containing your right thumb print) to a paper towel. Do the following in a fume hood or in a well ventilated area. The ethanol used in preparing the ninhydrin solution is volatile and flammable. Keep this solution away from open flames. Wear plastic gloves when working with the ninhydrin solution as it will react with the amino acids in your hand and turn them blue to purple!
- Dip the tip of the brush into the ninhydrin solution and carefully dab this liquid over the fingerprint area. Do not use too much pressure since that will destroy the print. Cotton wads held with tweezers can also be used to dab the liquid onto the fingerprint area.
- Allow the paper to dry. It may take 24 hrs to develop. Observe the print under a magnifying glass and compare with the fingerprint obtained above.
- If the print does not develop, expose the paper to the fumes from ammonia by opening a bottle of concentrated ammonia in the fume hood and holding the paper with the print over the opening of the bottle. Alternatively, a steam iron may be used.
Demonstration 4: Lipstick Fluorescence
Many substances contain molecules that absorb radiation in the ultraviolet portion of the spectrum and because of certain intramolecular phenomena, emit radiation in the visible region of the spectrum. This phenomenon is called fluorescence (see Photochemistry module). In this activity you will examine the fluorescence of lipstick. These observations suggest why makeup may appear different in a disco, or under a street light, as compared to inside a room.
Materials
Lipstick samples
Filter paper
Ultraviolet lamp
Safety
Ultraviolet light can damage eyes. Do not look directly at the ultraviolet light.
Procedure
- Obtain the several lipstick samples to be used in this demonstration. Record the color and manufacturer for each type, then describe the color.
- Use a sheet of filter paper. Place a smear of each kind of lipstick onto the paper, using a mark about one-half inch long. Write the brand or other identification next to the smear for identification.
- Expose the filter paper containing the lipstick stains to an ultraviolet lamp in a dark room. Observe which of the lipstick samples fluoresce under the light and circle those samples.
Demonstration 5: Examination of Hair
Hair is a common form of evidence in many homicide and sexual assault cases. Hair from any part of the body exhibits a range of characteristics such as color, length, and diameter. The parts of a hair that are easily seen by use of a microscope under magnification are the medulla and cortex. It is very difficult to see the hair cuticle. Figure 9 illustrates the parts of a hair and the various types of hair.
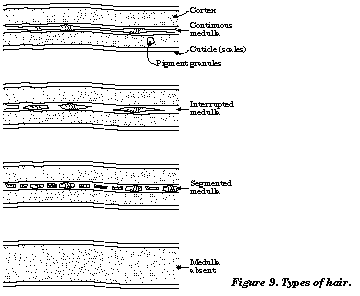
Many animal hairs are easily distinguished from human hairs by the size and shape of their medullae and the patterns of their cuticle or scale structures (see Figure 10). Synthetic fibers have no medulla or scalepattern andarethereforereadily distinguishable from animal hair.

Materials
Microscope slides
Compoundmicroscope(100X is a good magnification)
Tissue paper
Tweezers
Hairsample(yours,animal)
Fiber sample
Glycerol (glycerin)
Safety:
Glycerol (glycerin) should not be ingested.
Procedure
- Obtain a strand of human hair and place it on a microscope slide.
- Place a small drop of glycerol on the hair in order to hold it in place, and put a cover slip over it.
- Placethe slideonthestage ofthecompoundmicroscope, clipinplace,and adjust the magnification at 100X.
- Locate the root end of the hair, if it has one. If the hair has been forcibly pulled out, you will see a bulb-shaped enlargement. This is the root.
- Make a sketch of what you see.
- Scan along the length of the hair body. Is the medulla (center) fragmented (present in isolated spots), interrupted (long columns with open spaces now and then), or continuous (unbroken column)? Make a sketch of the medulla you observe from the hair sample.
- Note the color, diameter, and pigmentation of the hair.
- Examine the tip of the hair. This can be done by observing a gradual tapering of the hair. If the hair has been recently cut, you will see a square tip where the hair ends abruptly. Normally, hair tapers to a fine point as it grows. If hair has split-ends, it is normally due to artificial waving or bleaching, although repeated brushing may also produce this effect.
- Repeat Steps 1-8 for the sample of animal hair. Note any similarities and differences with the human hair.
- Try to obtain various colors of hair from other persons in the class. Make comparisons about similarities and differences.
- Obtain a fiber strand (cotton, nylon, silk, wool, Dacron, linen, rayon) and observe the color. If present, the color is due to a dye or stain. Prepare a microscope slide of the fiber and try to determine if the dye penetrates the fiber, or is found only on the surface.
- Examine other fiber characteristics such as diameter, whether the surface is rough or smooth, whether the fiber is twisted or straight, whether it is continuous, or segmented, whether it is round, flat, oval, or has some other shape.
- Compare and contrast human hair, animal hair, and fiber.
Counterintuitive Examples and Discrepant Events
Metaphors and Analogies
| TABLE OF CONTENTS | TOPIC OVERVIEW | CONCEPT/SKILLS DEVELOPMENT | LINKS/CONNECTIONS | EXTENSIONS |
|---|